Synthesis of 1-Substituted Bicyclo[2.1.1]hexan-2-ones via a Sequential SmI2-Mediated Pinacol Coupling and Acid-Catalyzed Pinacol Rearrangement Reaction.
IF 4.9
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A two-step procedure, combining a SmI2-mediated transannular pinacol coupling reaction with an acid-catalyzed pinacol rearrangement process, was employed to prepare a diverse range of 1-substituted bicyclo[2.1.1]hexan-5-ones from cyclobutanedione derivatives. To underscore the significance of these bicyclic ketones in drug synthesis, an sp3-rich analog of nitazoxanide, a well-known antiparasitic and antiviral agent, was synthesized.
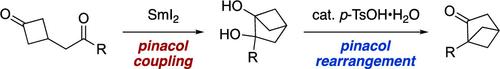
通过 SmI2 介导的频哪醇偶联反应和酸催化的频哪醇重排反应合成 1-取代的双环[2.1.1]己-2-酮。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


