Skeletal Editing of Mechanically Interlocked Molecules: Nitrogen Atom Deletion from Crown Ether-Dibenzylammonium Rotaxanes
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
Removing the nitrogen atom from secondary amines while simultaneously linking the remaining fragments is a powerful form of late-stage skeletal editing. Here, we report its use for the deletion of the nitrogen atom of the dibenzylammonium template used to assemble crown ether rotaxanes. The reaction uses an anomeric amide that activates secondary amines to generate a carbon–carbon bond that replaces the amine nitrogen. Despite the potential for dethreading of the intermediate diradical pair, the nitrogen atom was successfully deleted from a series of rotaxane axles as long as the macrocycle could access coconformations that did not inhibit the reaction of the amine group. The skeletally edited interlocked molecules were obtained directly from the parent crown ether-dibenzylammonium rotaxanes in modest yields (23–36%) and characterized by NMR spectroscopy, mass spectrometry, and X-ray crystallography. One skeletally edited rotaxane shows a network of weak CH···O hydrogen bonds between the crown ether and benzylic methylene groups of the axle in the solid state, in place of the crown ether-ammonium binding motif used to assemble the parent, unedited, rotaxane.
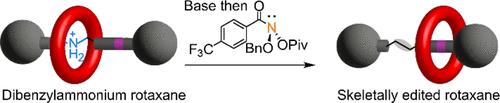
机械连锁分子的骨架编辑:冠醚-二苄基铵轮烷的氮原子删除
去除仲胺中的氮原子,同时连接剩余的片段是一种功能强大的后期骨架编辑形式。在此,我们报告了这种方法在去除用于组装冠醚轮烷的二苄基铵模板的氮原子时的应用。该反应使用活化仲胺的同分异构酰胺,生成取代胺氮的碳-碳键。尽管中间二叉对有可能发生拆线,但只要大环能够进入不抑制胺基反应的共构型,氮原子就能成功地从一系列轮烷轴中删除。从母体冠醚-二苄基铵轮烷中直接获得了骨架编辑的互锁分子,产率不高(23-36%),并通过核磁共振光谱、质谱和 X 射线晶体学对其进行了表征。其中一种经过骨架编辑的轮烷在固态下显示出冠醚与轴的苄基亚甲基之间的弱 CH-O 氢键网络,取代了用于组装未经编辑的母体轮烷的冠醚-铵结合基团。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


