Synthesis and evaluation of novel tetrahydroisoquinoline-benzo[h]chromen-4-one conjugates as dual ABCB1/CYP1B1 inhibitors for overcoming MDR in cancer
IF 3.3
3区 医学
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
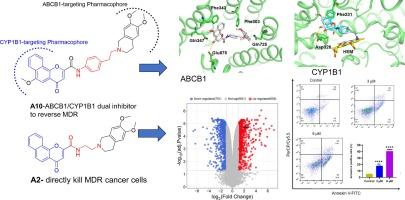
The emergence of multidrug resistance (MDR) in malignant tumors is one of the major threats encountered currently by many chemotherapeutic agents. Among the various mechanisms involved in drug resistance, P-glycoprotein (P-gp, ABCB1), a member of the ABC transporter family that significantly increases the efflux of various anticancer drugs from tumor cells, and the metabolic enzyme CYP1B1 are widely considered to be two critical targets for overcoming MDR. Unfortunately, no MDR modulator has been approved by the FDA to date. In this study, based on pharmacophore hybridization, bioisosteric and fragment-growing strategies, we designed and synthesized 11 novel tetrahydroisoquinoline-benzo[h]chromen-4-one conjugates as dual ABCB1/CYP1B1 inhibitors. Among them, the preferred compound A10 exhibited the best MDR reversal activity (IC50 = 0.25 μM, RF = 44.4) in SW620/AD300 cells, being comparable to one of the most potent third-generation P-gp inhibitors WK-X-34. In parallel, this dual ABCB1/CYP1B1 inhibitory effect drives compound A10 exhibiting prominent drug resistance reversal activity to doxorubicin (IC50 = 4.7 μM, RF = 13.7) in ABCB1/CYP1B1-overexpressing DOX-SW620/AD300-1B1 resistant cells, which is more potent than that of the CYP1B1 inhibitor ANF. Furthermore, although compound A2 possessed moderate ABCB1/CYP1B1 inhibitory activity, it showed considerable antiproliferative activity towards drug-resistant SW620/AD300 and MKN45-DDP-R cells, which may be partly related to the increase of PUMA expression to promote the apoptosis of the drug-resistant MKN45-DDP-R cells as confirmed by proteomics and western blot assay. These results indicated that the tetrahydroisoquinoline-benzo[h]chromen-4-one conjugates may provide a fundamental scaffold reference for further discovery of MDR reversal agents.
合成和评估新型四氢异喹啉-苯并[h]色烯-4-酮共轭物作为 ABCB1/CYP1B1 双重抑制剂用于克服癌症的 MDR
恶性肿瘤中出现的多药耐药性(MDR)是许多化疗药物目前面临的主要威胁之一。在导致耐药性的各种机制中,P-糖蛋白(P-gp,ABCB1)和代谢酶 CYP1B1 被广泛认为是克服 MDR 的两个关键靶点,P-糖蛋白(P-gp,ABCB1)是 ABC 转运体家族的成员,能显著增加各种抗癌药物从肿瘤细胞中的外流。遗憾的是,迄今为止还没有一种 MDR 调节剂获得 FDA 批准。在本研究中,我们基于药效杂交、生物异构和片段生长策略,设计并合成了 11 种新型四氢异喹啉-苯并[h]色烯-4-酮共轭物,作为 ABCB1/CYP1B1 双重抑制剂。其中,首选化合物 A10 在 SW620/AD300 细胞中表现出最佳的 MDR 逆转活性(IC50 = 0.25 μM,RF = 44.4),可与最有效的第三代 Pp 抑制剂之一 WK-X-34 相媲美。与此同时,这种 ABCB1/CYP1B1 双重抑制作用促使化合物 A10 在 ABCB1/CYP1B1 表达缺失的 DOX-SW620/AD300-1B1 耐药细胞中对多柔比星具有显著的耐药逆转活性(IC50 = 4.7 μM,RF = 13.7),比 CYP1B1 抑制剂 ANF 更强。此外,虽然化合物 A2 具有中等的 ABCB1/CYP1B1 抑制活性,但它对耐药的 SW620/AD300 和 MKN45-DDP-R 细胞却具有相当强的抗增殖活性,这可能与蛋白质组学和 Western 印迹检测证实的 PUMA 表达增加促进耐药的 MKN45-DDP-R 细胞凋亡有部分关系。这些结果表明,四氢异喹啉-苯并[h]色烯-4-酮共轭物可为进一步发现 MDR 逆转剂提供基本的支架参考。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Bioorganic & Medicinal Chemistry
医学-生化与分子生物学
CiteScore
6.80
自引率
2.90%
发文量
413
审稿时长
17 days
期刊介绍:
Bioorganic & Medicinal Chemistry provides an international forum for the publication of full original research papers and critical reviews on molecular interactions in key biological targets such as receptors, channels, enzymes, nucleotides, lipids and saccharides.
The aim of the journal is to promote a better understanding at the molecular level of life processes, and living organisms, as well as the interaction of these with chemical agents. A special feature will be that colour illustrations will be reproduced at no charge to the author, provided that the Editor agrees that colour is essential to the information content of the illustration in question.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


