Synthesis of (−)-Monanchoradin A and (−)-Crambescin A2 392 Based on a Cyclization–Carbonylation–Cyclization Cascade
IF 4.9
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Syntheses of guanidino alkaloids (−)-monanchoradin A and (−)-crambescin A2 392 are described. The key feature of the syntheses is the cyclization–carbonylation–cyclization cascade of the optically active propargyl guanidine. The bicyclic guanidino cores bearing an asymmetric center and ester or carboxylic acid functionality were constructed in a single step. The carboxylic acid was then converted to (−)-monanchoradin A and (−)-crambescin A2 392.
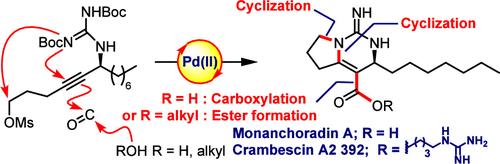
基于环化-羰基化-环化级联的 (-)-Monanchoradin A 和 (-)-Crambescin A2 的合成 392
介绍了胍类生物碱 (-)-monanchoradin A 和 (-)-crambescin A2 392 的合成。合成的主要特征是光学活性丙炔基胍的环化-羰基化-环化级联反应。带有不对称中心和酯或羧酸官能团的双环胍基核只需一步就能合成。然后将羧酸转化为 (-)-monanchoradin A 和 (-)-crambescin A2 392。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


