Construction of cytochrome P450 3A and P-glycoprotein knockout rats with application in rivaroxaban-verapamil interactions
IF 5.3
2区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Cytochrome P450 3A (CYP3A) and P-glycoprotein (P-gp), as important metabolic enzymes and transporters, participate in the biological transformation and transport of many substances in the body. CYP3A and P-gp are closely related, with very high substrate overlap and regulation similarity, making it particularly difficult to investigate the function of one or the other individually in vivo. Rivaroxaban and verapamil are commonly used together to treat nonvalvular atrial fibrillation in clinical practice. However, this combination therapy can increase systemic exposure to rivaroxaban and the risk of major bleeding and intracranial hemorrhage. In this study, Cyp3a1/2 and Mdr1a/b quadruple gene knockout (qKO) rat model was generated and characterized for the first time. CYP3A1/2 and P-gp are completely absent in this novel rat model. Then, the qKO rat model was applied for the evaluation of the drug-drug interactions (DDI) between rivaroxaban and verapamil. The results demonstrated that CYP3A and P-gp were jointly and selectively involved in the pharmacokinetic interactions between rivaroxaban and verapamil. This study may provide useful information for understanding the role of CYP3A and P-gp in rivaroxaban-verapamil therapy and predicting the potential interaction between CYP3A and P-gp.
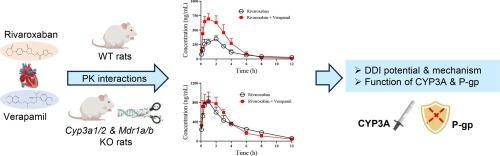
构建细胞色素 P450 3A 和 P-glycoprotein 基因敲除大鼠,并将其应用于利伐沙班与维拉帕米的相互作用。
细胞色素 P450 3A(CYP3A)和 P 糖蛋白(P-gp)作为重要的代谢酶和转运体,参与了许多物质在体内的生物转化和转运。CYP3A 和 P-gp 关系密切,底物重叠和调控相似度极高,因此在体内单独研究二者的功能尤为困难。在临床实践中,利伐沙班和维拉帕米通常一起用于治疗非瓣膜性心房颤动。然而,这种联合疗法会增加利伐沙班的全身暴露,增加大出血和颅内出血的风险。本研究首次建立了Cyp3a1/2和Mdr1a/b四重基因敲除(qKO)大鼠模型,并对其进行了表征。在这种新型大鼠模型中,CYP3A1/2 和 P-gp 基因完全缺失。然后,将 qKO 大鼠模型用于评估利伐沙班和维拉帕米之间的药物相互作用(DDI)。结果表明,CYP3A和P-gp共同选择性地参与了利伐沙班和维拉帕米之间的药代动力学相互作用。这项研究可为了解 CYP3A 和 P-gp 在利伐沙班-维拉帕米治疗中的作用以及预测 CYP3A 和 P-gp 之间的潜在相互作用提供有用信息。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Biochemical pharmacology
医学-药学
CiteScore
10.30
自引率
1.70%
发文量
420
审稿时长
17 days
期刊介绍:
Biochemical Pharmacology publishes original research findings, Commentaries and review articles related to the elucidation of cellular and tissue function(s) at the biochemical and molecular levels, the modification of cellular phenotype(s) by genetic, transcriptional/translational or drug/compound-induced modifications, as well as the pharmacodynamics and pharmacokinetics of xenobiotics and drugs, the latter including both small molecules and biologics.
The journal''s target audience includes scientists engaged in the identification and study of the mechanisms of action of xenobiotics, biologics and drugs and in the drug discovery and development process.
All areas of cellular biology and cellular, tissue/organ and whole animal pharmacology fall within the scope of the journal. Drug classes covered include anti-infectives, anti-inflammatory agents, chemotherapeutics, cardiovascular, endocrinological, immunological, metabolic, neurological and psychiatric drugs, as well as research on drug metabolism and kinetics. While medicinal chemistry is a topic of complimentary interest, manuscripts in this area must contain sufficient biological data to characterize pharmacologically the compounds reported. Submissions describing work focused predominately on chemical synthesis and molecular modeling will not be considered for review.
While particular emphasis is placed on reporting the results of molecular and biochemical studies, research involving the use of tissue and animal models of human pathophysiology and toxicology is of interest to the extent that it helps define drug mechanisms of action, safety and efficacy.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


