Integrated transcriptomic and proteomic analysis revealed the regulatory role of 5-azacytidine in kenaf salt stress alleviation
IF 2.8
2区 生物学
Q2 BIOCHEMICAL RESEARCH METHODS
引用次数: 0
Abstract
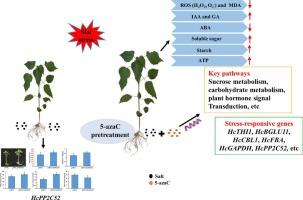
Salinity stress limits agricultural production. The DNA methyltransferase inhibitor, 5-azacitidine (5-azaC), plays a role in plant abiotic stress regulation, but its molecular basis in mediating salinity tolerance in kenaf remains unclear. To investigate the effects on 5-azaC on alleviating salt stress, kenaf seedlings were pre-treated with 0, 50, 100, 150, and 200 μM 5-azaC and then exposed to 150 mM NaCl in a nutrient solution. Physiological, transcriptomic, and proteomic analyses were conducted on the root system to understand the regulatory mechanism of 5-azaC (comparing 5-azaC150 and control group 5-azaC0) under salt stress. The results indicated that 5-azaC significantly mitigated salt stress in kenaf by activating the antioxidant system, reducing reactive oxygen species (ROS), and increasing starch, soluble sugars, and adenosine triphosphate (ATP) content. A total of 14,348 differentially expressed genes (DEGs) and 313 differentially abundant proteins (DAPs) were identified. Combined proteomic and transcriptomic analysis revealed 27 DEGs/DAPs, with jointly up-regulated proteins (genes) including HcTHI1, HcBGLU11, and HcCBL1, and jointly down-regulated proteins (genes) including HcGAPDH, HcSS, and HcPP2C52. Overexpression and virus-induced gene silencing (VIGS) of HcPP2C52 demonstrated its role as a negative regulator of salt tolerance. These findings provide insights into the regulatory role of 5-azaC in plant responses to abiotic stresses.
Significance
The specific molecular mechanism by which 5-azaC affects gene expression and protein activity of kenaf has been revealed, leading to enhanced salt tolerance.
转录组和蛋白质组的综合分析揭示了 5-azacytidine 在减轻剑麻盐胁迫中的调控作用。
盐分胁迫限制了农业生产。DNA 甲基转移酶抑制剂--5-氮杂胞苷(5-azaC)在植物非生物胁迫调控中发挥作用,但其介导剑麻耐盐性的分子基础仍不清楚。为了研究 5-azaC 对缓解盐胁迫的影响,我们用 0、50、100、150 和 200 μM 5-azaC 预处理剑麻幼苗,然后将其置于 150 mM NaCl 营养液中。对根系进行生理、转录组和蛋白质组分析,以了解盐胁迫下 5-azaC 的调控机制(5-azaC150 与对照组 5-azaC0比较)。结果表明,5-azaC 能激活抗氧化系统,减少活性氧(ROS),增加淀粉、可溶性糖和三磷酸腺苷(ATP)的含量,从而显著缓解剑麻的盐胁迫。共鉴定出 14 348 个差异表达基因(DEG)和 313 个差异丰富蛋白(DAP)。结合蛋白质组和转录组分析发现了 27 个 DEGs/DAPs,共同上调的蛋白质(基因)包括 HcTHI1、HcBGLU11 和 HcCBL1,共同下调的蛋白质(基因)包括 HcGAPDH、HcSS 和 HcPP2C52。HcPP2C52的过表达和病毒诱导基因沉默(VIGS)证明了其作为耐盐性负调控因子的作用。这些发现深入揭示了 5-azaC 在植物对非生物胁迫反应中的调控作用。意义:揭示了 5-azaC 影响剑麻基因表达和蛋白质活性的具体分子机制,从而增强了剑麻的耐盐性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of proteomics
生物-生化研究方法
CiteScore
7.10
自引率
3.00%
发文量
227
审稿时长
73 days
期刊介绍:
Journal of Proteomics is aimed at protein scientists and analytical chemists in the field of proteomics, biomarker discovery, protein analytics, plant proteomics, microbial and animal proteomics, human studies, tissue imaging by mass spectrometry, non-conventional and non-model organism proteomics, and protein bioinformatics. The journal welcomes papers in new and upcoming areas such as metabolomics, genomics, systems biology, toxicogenomics, pharmacoproteomics.
Journal of Proteomics unifies both fundamental scientists and clinicians, and includes translational research. Suggestions for reviews, webinars and thematic issues are welcome.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


