Computational Approach to the Study of Acidochromic Properties of Donor-π-Acceptor Systems Based on Dimethylamino-Substituted Dyes With Intramolecular Charge Transfer
Abstract
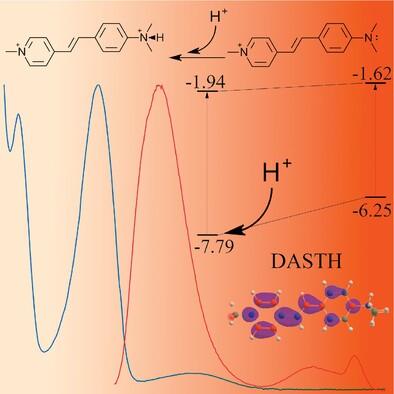
This work is devoted to the study of the influence of protonation on the photophysical properties of dimethylamino-substituted styryl dyes. The formation of a bond on the unshared nitrogen electron pair involved in conjugation with the dye chromophore changes the mobility of the terminal group of the donor fragment and thereby “switches” the molecule from n-π* to π-π* mode. The correlations found between changes in the electron density of the dye in native form and during protonation and changes in its properties contribute to the study of the ground and excited states of these compounds and the energy transitions between them. Comparison with analog compounds and consideration of vibronic effects allow us to evaluate the potential advantages and limitations of the TD-DFT method in the calculation of electronic transitions in styryl dyes. The work contributes to the understanding of the influence of protonation on the behavior of dyes with a nitrogen atom in the donor part of the chromophore of the molecule. The patterns found can be applied to similar chromophore systems.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


