Ab Initio Molecular Dynamics Approach for Oxidation of SiC Surface in Contact with Aqueous H2O2 Solutions
IF 3.3
3区 化学
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
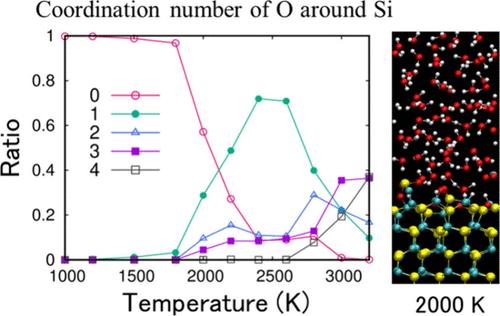
Controlling the oxidation of silicon carbide (SiC) is a key factor in fabricating SiC-based devices, such as power-integrated circuits and thermal protection systems. Oxidation is particularly utilized for machining SiC in conjunction with chemical–mechanical polishing. However, a thorough understanding of SiC oxidation at the microscopic level is lacking. Here, we performed ab initio molecular dynamics simulations to elucidate the microscopic mechanisms of the oxidation of OH- and H-terminated SiC surfaces in contact with an oxidizing solution─aqueous H2O2 solution. We found that the OH-terminated (0001) C-face surface of SiC is more easily oxidized than the OH-terminated (0001) Si-face and H-terminated C- and Si-faces. The stability of the C–O and Si–O species on the surface plays a key role in the oxidation of the OH-terminated surfaces. The effect of H2O2 concentration on the oxidation of the C-face was also examined. We found that the OH radicals generated from H2O2 molecules undergo a facile conversion to H2O molecules in a pure H2O2 solution. This phenomenon may explain the counterintuitive dependence of oxidation on the H2O2 concentration observed in the experiments.
碳化硅表面与 H2O2 水溶液接触氧化的 Ab Initio 分子动力学方法
控制碳化硅(SiC)的氧化是制造基于碳化硅的设备(如功率集成电路和热保护系统)的关键因素。氧化尤其适用于结合化学机械抛光加工碳化硅。然而,目前还缺乏对碳化硅微观氧化的透彻了解。在此,我们进行了 ab initio 分子动力学模拟,以阐明与氧化溶液(H2O2 水溶液)接触的 OH 端和 H 端碳化硅表面氧化的微观机制。我们发现,SiC 的 OH 端接(0001)C 面比 OH 端接(0001)Si 面以及 H 端接 C 面和 Si 面更容易被氧化。表面上 C-O 和 Si-O 物种的稳定性在 OH 端接表面的氧化过程中起着关键作用。我们还研究了 H2O2 浓度对 C 面氧化的影响。我们发现,在纯 H2O2 溶液中,H2O2 分子产生的 OH 自由基很容易转化为 H2O 分子。这一现象可能解释了实验中观察到的氧化作用对 H2O2 浓度的反直觉依赖性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

The Journal of Physical Chemistry C
化学-材料科学:综合
CiteScore
6.50
自引率
8.10%
发文量
2047
审稿时长
1.8 months
期刊介绍:
The Journal of Physical Chemistry A/B/C is devoted to reporting new and original experimental and theoretical basic research of interest to physical chemists, biophysical chemists, and chemical physicists.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


