Assessing the diagnostic utility of tRNA-derived fragments as biomarkers of head and neck cancer
IF 5
2区 医学
Q2 Medicine
引用次数: 0
Abstract
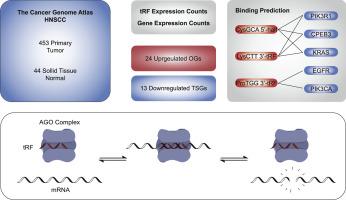
Roughly 54,000 individuals are diagnosed with head and neck cancers in the United States yearly. Transfer RNA-derived fragments (tRF) are the products of enzymatic cleavage of precursor tRNAs, and have been proposed for use as biomarkers of head and neck cancer. In this study, we aim to further analyze the utility that tRFs might provide as biomarkers of head and neck cancer. tRF read counts were obtained for 453 tumor and 44 adjacent normal tissue samples and used to construct a gradient boosting diagnostic model. Although we identified 129 tRFs that were significantly dysregulated between these samples, the model achieved a sensitivity of only 69 % and a specificity of 59 %. tRFs are thought to induce the degradation of mRNA transcripts containing a complementary “seed” region. Despite the above performances, we chose to explore this concept of translational regulation by analyzing these tRFs for inverse correlation to the expression of select oncogenes and tumor suppressor genes implicated in head and neck cancer. Among others, CysGCA 5′-half and LysCTT 3′-tRF were upregulated in the tumor samples, and corresponded to decreased expression of PIK3R1, AKT1, and CPEB3. These transcripts were further found to contain numerous significantly complementary sites at which tRF-mediated mRNA degradation might occur. Although these tRFs did appear to correlate to many of the oncogenic metrics analyzed, we believe that additional research is needed before they might be used to improve the diagnosis, treatment, and survival of patients with this disease.
评估 tRNA 衍生片段作为头颈癌生物标记物的诊断效用
在美国,每年约有 54,000 人被诊断出患有头颈部癌症。转运核糖核酸衍生片段(tRF)是前体 tRNA 酶解的产物,已被建议用作头颈部癌症的生物标志物。在这项研究中,我们旨在进一步分析 tRF 作为头颈癌生物标志物的效用。我们获得了 453 份肿瘤样本和 44 份邻近正常组织样本的 tRF 读数,并将其用于构建梯度增强诊断模型。虽然我们发现了 129 个在这些样本之间明显失调的 tRFs,但该模型的灵敏度仅为 69%,特异度为 59%。tRFs 被认为会诱导含有互补 "种子 "区域的 mRNA 转录本降解。尽管有上述表现,我们还是选择通过分析这些 tRFs 与头颈部癌症中某些癌基因和抑癌基因表达的反相关性来探索这一翻译调控概念。其中,CysGCA 5′-half和LysCTT 3′-tRF在肿瘤样本中上调,并与PIK3R1、AKT1和CPEB3的表达下降相对应。研究进一步发现,这些转录本包含许多显著互补的位点,tRF 介导的 mRNA 降解可能就发生在这些位点上。虽然这些 tRFs 似乎与许多分析的致癌指标相关,但我们认为还需要进行更多的研究,才能将它们用于改善这种疾病患者的诊断、治疗和生存。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Translational Oncology
ONCOLOGY-
CiteScore
8.40
自引率
2.00%
发文量
314
审稿时长
54 days
期刊介绍:
Translational Oncology publishes the results of novel research investigations which bridge the laboratory and clinical settings including risk assessment, cellular and molecular characterization, prevention, detection, diagnosis and treatment of human cancers with the overall goal of improving the clinical care of oncology patients. Translational Oncology will publish laboratory studies of novel therapeutic interventions as well as clinical trials which evaluate new treatment paradigms for cancer. Peer reviewed manuscript types include Original Reports, Reviews and Editorials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


