Ablating the glutaredoxin-2 (Glrx2) gene protects male mice against non-alcoholic fatty liver disease (NAFLD) by limiting oxidative distress
IF 7.1
2区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
In the present study, we investigated the consequences of deleting the glutaredoxin-2 gene (Glrx2−/−) on the development of non-alcoholic fatty liver disease (NAFLD) in male and female C57BL6N mice fed a control (CD) or high-fat diet (HFD). We report that the HFD induced a significant increase in body mass in the wild-type (Wt) and Glrx2−/− male, but not female, mice, which was associated with the hypertrophying of the abdominal fat. Interestingly, while the Wt male mice fed the HFD developed NAFLD, the deletion of the Glrx2 gene mitigated vesicle formation, intrahepatic lipid accumulation, and fibrosis in the males. The protective effect associated with ablating the Glrx2 gene in male mice was due to enhancement of mitochondrial redox buffering capacity. Specifically, liver mitochondria from male Glrx2−/− fed a CD or HFD produced significantly less hydrogen peroxide (mtH2O2), had lower malondialdehyde levels, greater activities for glutathione peroxidase and thioredoxin reductase, and less protein glutathione mixed disulfides (PSSG) when compared to the Wt male mice fed the HFD. These effects correlated with the S-glutathionylation of α-ketoglutarate dehydrogenase (KGDH), a potent mtH2O2 source and key redox sensor in hepatic mitochondria. In comparison to the male mice, both Wt and Glrx2−/− female mice displayed almost complete resistance to HFD-induced body mass increases and the development of NAFLD, which was attributed to the superior redox buffering capacity of the liver mitochondria. Together, our findings show that modulation of mitochondrial S-glutathionylation signaling through Glrx2 augments resistance of male mice towards the development of NAFLD through preservation of mitochondrial redox buffering capacity. Additionally, our findings demonstrate the sex dimorphisms associated with the manifestation of NAFLD is related to the superior redox buffering capacity and modulation of the S-glutathionylome in hepatic mitochondria from female mice.
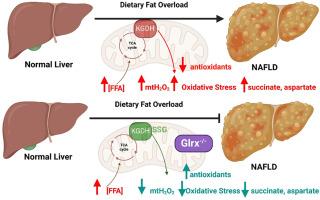
消减谷胱甘肽-2(Glrx2)基因可通过限制氧化损伤保护雄性小鼠免受非酒精性脂肪肝(NAFLD)的侵害。
在本研究中,我们研究了删除谷拉德酵素-2基因(Glrx2-/-)对以对照组(CD)或高脂饮食(HFD)喂养的雌雄C57BL6N小鼠非酒精性脂肪肝(NAFLD)发生的影响。我们报告说,高脂饮食诱导野生型(Wt)和Glrx2-/-雄性小鼠(而非雌性)的体重显著增加,这与腹部脂肪肥厚有关。有趣的是,喂食高密度脂蛋白胆固醇(HFD)的Wt雄性小鼠出现了非酒精性脂肪肝,而删除Glrx2基因则可减轻雄性小鼠体内囊泡的形成、肝内脂质积累和纤维化。在雄性小鼠中删除 Glrx2 基因所产生的保护作用是由于线粒体氧化还原缓冲能力的增强。具体来说,与喂食高氟日粮的 Wt 雄性小鼠相比,喂食 CD 或高氟日粮的 Glrx2-/-雄性小鼠肝脏线粒体产生的过氧化氢(mtH2O2)明显减少,丙二醛水平降低,谷胱甘肽过氧化物酶和硫代还原酶活性提高,蛋白质谷胱甘肽混合二硫化物(PSSG)减少。这些影响与α-酮戊二酸脱氢酶(KGDH)的S-谷胱甘肽化有关,KGDH是肝线粒体中一个有效的mtH2O2来源和关键的氧化还原传感器。与雄性小鼠相比,Wt和Glrx2-/-雌性小鼠对HFD诱导的体重增加和非酒精性脂肪肝的发展表现出几乎完全的抵抗力,这归因于肝线粒体卓越的氧化还原缓冲能力。总之,我们的研究结果表明,通过Glrx2调节线粒体S-谷胱甘肽化信号传导,可通过保护线粒体氧化还原缓冲能力增强雄性小鼠对非酒精性脂肪肝发生的抵抗力。此外,我们的研究结果表明,与非酒精性脂肪肝表现相关的性别二态性与雌性小鼠肝线粒体的卓越氧化还原缓冲能力和S-谷胱甘肽组的调节有关。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Free Radical Biology and Medicine
医学-内分泌学与代谢
CiteScore
14.00
自引率
4.10%
发文量
850
审稿时长
22 days
期刊介绍:
Free Radical Biology and Medicine is a leading journal in the field of redox biology, which is the study of the role of reactive oxygen species (ROS) and other oxidizing agents in biological systems. The journal serves as a premier forum for publishing innovative and groundbreaking research that explores the redox biology of health and disease, covering a wide range of topics and disciplines. Free Radical Biology and Medicine also commissions Special Issues that highlight recent advances in both basic and clinical research, with a particular emphasis on the mechanisms underlying altered metabolism and redox signaling. These Special Issues aim to provide a focused platform for the latest research in the field, fostering collaboration and knowledge exchange among researchers and clinicians.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


