Simultaneous enhancement of cellular and humoral immunity by the lymph node-targeted cholesterolized TLR7 agonist liposomes
IF 14.7
1区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Toll-like receptor (TLR) agonists, as promising adjuvants and immunotherapeutic agents, have the potential to enhance immune responses and modulate antigen-dependent T-cell immune memory through activation of distinct signaling pathways. However, their clinical application is hindered by uncontrolled systemic inflammatory reactions. Therefore, it is imperative to create a vaccine adjuvant for TLR receptors that ensures both safety and efficacy. In this study, we designed lymph node-targeted cholesterolized TLR7 agonist cationic liposomes (1V209-Cho-Lip+) to mitigate undesired side effects. Co-delivery of the model antigen OVA and cholesterolized TLR7 agonist facilitated DC maturation through TLR activation while ensuring optimal presentation of the antigen to CD8+ T cells. The main aim of the present study is to evaluate the adjuvant effectiveness of 1V209-Cho-Lip+ in tumor vaccines. Following immunization with 1V209-Cho-Lip++OVA, we observed a pronounced "depot effect" and enhanced trafficking to secondary lymphoid organs. Prophylactic vaccination with 1V209-Cho-Lip++OVA significantly delays tumor development, prolongs mouse survival, and establishes durable immunity against tumor recurrence. Additionally, 1V209-Cho-Lip++OVA, while used therapeutic tumor vaccine, has demonstrated its efficacy in inhibiting tumor progression, and when combined with anti-PD-1, it further enhances antitumor effects. Therefore, the co-delivery of antigen and lymph node-targeted cholesterolized TLR7 agonist shows great promise as a cancer vaccine.
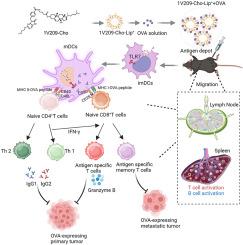
淋巴结靶向胆固醇化 TLR7 激动剂脂质体可同时增强细胞和体液免疫力
Toll 样受体(TLR)激动剂是一种很有前景的佐剂和免疫治疗剂,有可能通过激活不同的信号通路来增强免疫反应和调节抗原依赖性 T 细胞免疫记忆。然而,它们在临床上的应用却受到不受控制的全身性炎症反应的阻碍。因此,当务之急是为 TLR 受体创造一种既安全又有效的疫苗佐剂。在本研究中,我们设计了淋巴结靶向胆固醇化 TLR7 激动剂阳离子脂质体(1V209-Cho-Lip),以减轻不良副作用。模型抗原 OVA 和胆固醇化 TLR7 激动剂的联合给药有助于通过 TLR 激活促进 DC 成熟,同时确保抗原以最佳方式呈现给 CD8 T 细胞。本研究的主要目的是评估 1V209-Cho-Lip 在肿瘤疫苗中的佐剂效果。在使用 1V209-Cho-Lip+OVA 进行免疫接种后,我们观察到了明显的 "储藏效应 "和向次级淋巴器官的迁移增强。预防性接种 1V209-Cho-Lip+OVA 能显著延缓肿瘤的发展,延长小鼠的存活时间,并建立起防止肿瘤复发的持久免疫力。此外,1V209-Cho-Lip+OVA 作为治疗性肿瘤疫苗,在抑制肿瘤进展方面的疗效已得到证实,与抗-PD-1 结合使用时,可进一步增强抗肿瘤效果。因此,将抗原和淋巴结靶向胆固醇化 TLR7 激动剂联合递送作为癌症疫苗大有可为。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta Pharmaceutica Sinica. B
Pharmacology, Toxicology and Pharmaceutics-General Pharmacology, Toxicology and Pharmaceutics
CiteScore
22.40
自引率
5.50%
发文量
1051
审稿时长
19 weeks
期刊介绍:
The Journal of the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association oversees the peer review process for Acta Pharmaceutica Sinica. B (APSB).
Published monthly in English, APSB is dedicated to disseminating significant original research articles, rapid communications, and high-quality reviews that highlight recent advances across various pharmaceutical sciences domains. These encompass pharmacology, pharmaceutics, medicinal chemistry, natural products, pharmacognosy, pharmaceutical analysis, and pharmacokinetics.
A part of the Acta Pharmaceutica Sinica series, established in 1953 and indexed in prominent databases like Chemical Abstracts, Index Medicus, SciFinder Scholar, Biological Abstracts, International Pharmaceutical Abstracts, Cambridge Scientific Abstracts, and Current Bibliography on Science and Technology, APSB is sponsored by the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association. Its production and hosting are facilitated by Elsevier B.V. This collaborative effort ensures APSB's commitment to delivering valuable contributions to the pharmaceutical sciences community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


