Covalent Probes To Capture Legionella pneumophila Dup Effector Enzymes
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
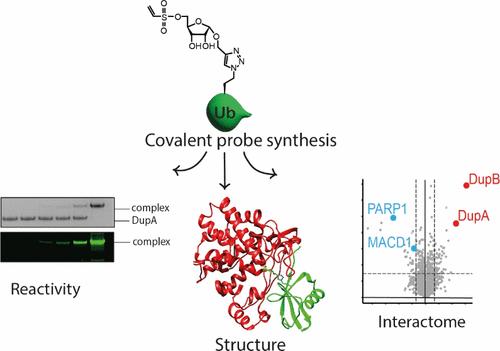
Upon infection of host cells, Legionella pneumophila releases a multitude of effector enzymes into the cell's cytoplasm that hijack a plethora of cellular activities, including the host ubiquitination pathways. Effectors belonging to the SidE-family are involved in noncanonical serine phosphoribosyl ubiquitination of host substrate proteins contributing to the formation of a Legionella-containing vacuole that is crucial in the onset of Legionnaires’ disease. This dynamic process is reversed by effectors called Dups that hydrolyze the phosphodiester in the phosphoribosyl ubiquitinated protein. We installed reactive warheads on chemically prepared ribosylated ubiquitin to generate a set of probes targeting these Legionella enzymes. In vitro tests on recombinant DupA revealed that a vinyl sulfonate warhead was most efficient in covalent complex formation. Mutagenesis and X-ray crystallography approaches were used to identify the site of covalent cross-linking to be an allosteric cysteine residue. The subsequent application of this probe highlights the potential to selectively enrich the Dup enzymes from Legionella-infected cell lysates.
捕捉嗜肺军团菌二价效应酶的共价探针
感染宿主细胞后,嗜肺军团菌会向细胞胞质释放大量效应酶,这些效应酶会劫持大量细胞活动,包括宿主泛素化途径。属于 SidE 家族的效应酶参与了宿主底物蛋白质的非规范丝氨酸磷酸核糖泛素化,有助于形成一个含军团菌的空泡,这对军团病的发病至关重要。这种动态过程会被称为 Dups 的效应器逆转,Dups 会水解磷酸核糖泛素化蛋白质中的磷酸二酯。我们在化学制备的核糖核苷泛素上安装了反应弹头,以生成一套针对这些军团菌酶的探针。对重组 DupA 的体外测试表明,乙烯基磺酸盐弹头形成共价复合物的效率最高。通过突变和 X 射线晶体学方法,确定了共价交联的位点是一个异位半胱氨酸残基。这种探针的后续应用凸显了从军团菌感染的细胞裂解物中选择性富集 Dup 酶的潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


