Polarized ATR-FTIR studies of DPPC/DPPG lipid bilayers doped with PLL
Abstract
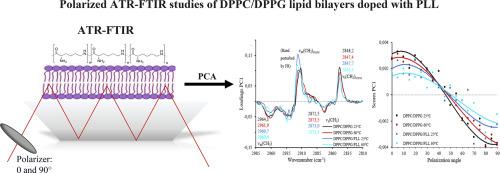
The structural characterization of lipid bilayers is crucial for understanding various biological processes and designing effective systems of drugs. Attenuated Total Reflection Fourier Transformed Infrared (ATR-FTIR) spectroscopy is a powerful technique for probing lipid membrane properties, but conventional methods face challenges in discerning molecular orientation and intermolecular interactions. The polarized ATR-FTIR spectroscopy combined with Principal Component Analysis (PCA) was used to investigate the interactions between positively charged peptide (poly-L-lysine (PLL)) and anionic lipid bilayers (dipalmitoylphosphatidylcholine/dipalmitoylphosphatidylglycerol, DPPC/DPPG). This integrated approach offers deeper insight into mutual interactions between proteins and lipids of membrane, providing a comprehensive understanding of structure of both protein and lipid bilayers components. The findings of this study shed light on the orientational order of lipid molecules in DPPC/DPPG membrane structure as well as secondary strictures of PPL peptides. It was shown that the presence of PLL peptide disrupts lipid molecule ordering within the lipid bilayer, primarily through electrostatic and H-bond interactions. The utility of polarized ATR-FTIR spectroscopy supported by PCA calculations in elucidating molecular organizations within complex membrane systems is emphasized.

| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


