Chiral Macrocycles for Enantioselective Recognition
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
The efficient synthesis of chiral macrocycles with highly enantioselective recognition remains a challenge. We have addressed this issue by synthesizing a pair of chiral macrocycles, namely, R/S-BINOL[2], achieving total isolated yields of up to 62% through a two-step reaction sequence. These macrocycles are readily purified by column chromatography over silica gel without the need for chiral separation, thus streamlining the overall synthesis. R/S-BINOL[2] demonstrated enantioselective recognition toward chiral ammonium salts, with enantioselectivity (KS/KR) values reaching up to 13.2, although less favorable separations were seen for other substrates. R/S-BINOL[2] also displays blue circularly polarized luminescence with a |glum| value of up to 2.2 × 10–3. The R/S-BINOL[2] macrocycles of this study are attractive as chiral hosts in that they both display enantioselective guest recognition and benefit from a concise, high-yielding synthesis. As such, they may have a role to play in chiral separations.
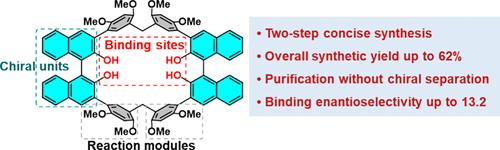
用于对映选择性识别的手性大环
高效合成具有高度对映选择性的手性大环仍然是一项挑战。针对这一问题,我们合成了一对手性大环,即 R/S-BINOL[2],通过两步反应顺序,总分离产率高达 62%。这些大环很容易通过硅胶柱层析法纯化,无需进行手性分离,从而简化了整个合成过程。R/S-BINOL[2] 对手性铵盐具有对映选择性识别能力,对映选择性(KS/KR)值高达 13.2,但对其他底物的分离效果较差。R/S-BINOL[2] 还显示出蓝色圆偏振发光,其|glum|值高达 2.2 × 10-3。本研究中的 R/S-BINOL[2] 大环作为手性宿主很有吸引力,因为它们既能显示对映选择性客体识别,又能受益于简便、高产的合成。因此,它们可以在手性分离中发挥作用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


