GOLM1 dictates acquired Lenvatinib resistance by a GOLM1-CSN5 positive feedback loop upon EGFR signaling activation in hepatocellular carcinoma
IF 6.9
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Lenvatinib is a multiple receptor tyrosine kinases inhibitor (TKI) authorized for first-line treatment of hepatocellular carcinoma (HCC). However, Lenvatinib resistance is common in HCC clinical treatment, highlighting the urgent need to understand mechanisms of resistance. Here, we identified Golgi membrane protein 1 (GOLM1), a type II transmembrane protein originally located in the Golgi apparatus, as a novel regulator of Lenvatinib resistance. We found GOLM1 was overexpressed in Lenvatinib resistant human HCC cell lines, blood and HCC samples. Additionally, GOLM1 overexpression contributes to Lenvatinib resistance and HCC progression in vitro and in vivo. Mechanistically, GOLM1 upregulates CSN5 expression through EGFR-STAT3 pathway. Reversely, CSN5 deubiquitinates and stabilizes GOLM1 protein by inhibiting ubiquitin-proteasome pathway of GOLM1. Furthermore, clinical specimens of HCC showed a positive correlation between the activation of the GOLM1-EGFR-STAT3-CSN5 axis. Finally, GOLM1 knockdown was found to act in synergy with Lenvatinib in subcutaneous and orthotopic mouse model. Overall, these findings identify a mechanism of resistance to Lenvatinib treatment for HCC, highlight an effective predictive biomarker of Lenvatinib response in HCC and show that targeting GOLM1 may improve the clinical benefit of Lenvatinib.
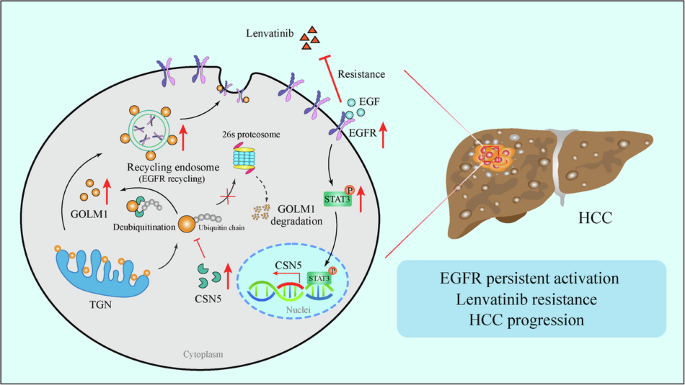
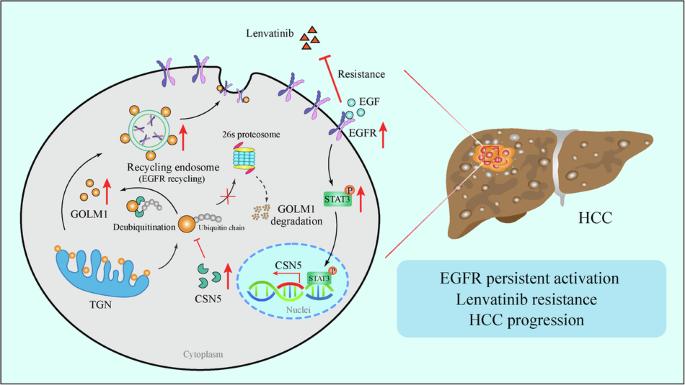
肝细胞癌中表皮生长因子受体(EGFR)信号激活后,GOLM1-CSN5正反馈环决定了伦伐替尼的耐药性
伦伐替尼是一种多受体酪氨酸激酶抑制剂(TKI),被授权用于肝细胞癌(HCC)的一线治疗。然而,伦伐替尼耐药在HCC临床治疗中很常见,这凸显了了解耐药机制的迫切需要。在这里,我们发现高尔基体膜蛋白1(GOLM1)是一种原位于高尔基体的II型跨膜蛋白,是伦伐替尼耐药性的新型调控因子。我们发现,GOLM1 在伦伐替尼耐药的人类 HCC 细胞系、血液和 HCC 样本中过表达。此外,GOLM1的过表达还有助于伦伐替尼耐药以及HCC在体外和体内的进展。从机理上讲,GOLM1通过表皮生长因子受体-STAT3通路上调CSN5的表达。反之,CSN5通过抑制GOLM1的泛素-蛋白酶体途径,去泛素并稳定GOLM1蛋白。此外,HCC 的临床标本显示,GOLM1-EGFR-STAT3-CSN5 轴的激活之间存在正相关。最后,在皮下和正位小鼠模型中发现,GOLM1 基因敲除与仑伐替尼具有协同作用。总之,这些研究结果确定了伦伐替尼治疗 HCC 的耐药机制,强调了伦伐替尼对 HCC 反应的有效预测生物标志物,并表明靶向 GOLM1 可提高伦伐替尼的临床疗效。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Oncogene
医学-生化与分子生物学
CiteScore
15.30
自引率
1.20%
发文量
404
审稿时长
1 months
期刊介绍:
Oncogene is dedicated to advancing our understanding of cancer processes through the publication of exceptional research. The journal seeks to disseminate work that challenges conventional theories and contributes to establishing new paradigms in the etio-pathogenesis, diagnosis, treatment, or prevention of cancers. Emphasis is placed on research shedding light on processes driving metastatic spread and providing crucial insights into cancer biology beyond existing knowledge.
Areas covered include the cellular and molecular biology of cancer, resistance to cancer therapies, and the development of improved approaches to enhance survival. Oncogene spans the spectrum of cancer biology, from fundamental and theoretical work to translational, applied, and clinical research, including early and late Phase clinical trials, particularly those with biologic and translational endpoints.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


