Linker-specific monoclonal antibodies present a simple and reliable detection method for scFv-based CARNK cells
IF 4.6
2区 医学
Q2 MEDICINE, RESEARCH & EXPERIMENTAL
Molecular Therapy-Methods & Clinical Development
Pub Date : 2024-08-22
DOI:10.1016/j.omtm.2024.101328
引用次数: 0
Abstract
Chimeric antigen receptor (CAR) T cell therapies have demonstrated significant successes in treating cancer. Currently, there are six approved CAR T cell products available on the market that target different malignancies of the B cell lineage. However, to overcome the limitations of CAR T cell therapies, other immune cells are being investigated for CAR-based cell therapies. CAR natural killer (NK) cells can be applied as allogeneic cell therapy, providing an economical, safe, and efficient alternative to autologous CAR T cells. To improve CAR research and future in-patient monitoring of cell therapeutics, a simple, reliable, and versatile CAR detection reagent is crucial. As most existing CARs contain a single-chain variable fragment (scFv) with either a Whitlow or a G4S linker site, linker-specific monoclonal antibodies (mAbs) can detect a broad range of CARs. This study demonstrates that these linker-specific mAbs can detect different CAR NK cells , spiked in whole blood, and within patient-derived tumor spheroids with high specificity and sensitivity, providing an effective and almost universal alternative for scFv-based CAR detection. Additionally, we confirm that linker-specific antibodies can be used for functional testing and enrichment of CAR NK cells, thereby providing a useful research tool to fast-track the development of novel CAR-based therapies.
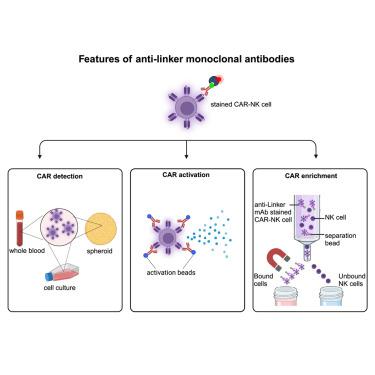
连接子特异性单克隆抗体为基于 scFv 的 CARNK 细胞提供了一种简单可靠的检测方法
嵌合抗原受体(CAR)T 细胞疗法在治疗癌症方面取得了巨大成功。目前,市场上有六种获批的 CAR T 细胞产品,针对 B 细胞系的不同恶性肿瘤。然而,为了克服 CAR T 细胞疗法的局限性,目前正在研究其他免疫细胞的 CAR 细胞疗法。CAR 自然杀伤(NK)细胞可用作异体细胞疗法,为自体 CAR T 细胞提供了一种经济、安全、高效的替代选择。为了改进 CAR 研究和未来对细胞疗法的住院监测,一种简单、可靠、多功能的 CAR 检测试剂至关重要。由于现有的大多数 CAR 都含有一个带有 Whitlow 或 G4S 连接位点的单链可变片段 (scFv),因此连接位点特异性单克隆抗体 (mAbs) 可以检测多种 CAR。本研究证明,这些连接子特异性 mAbs 能以高特异性和高灵敏度检测全血中的不同 CAR NK 细胞和患者来源的肿瘤球体内的 CAR NK 细胞,为基于 scFv 的 CAR 检测提供了一种有效且几乎通用的替代方法。此外,我们还证实了连接子特异性抗体可用于 CAR NK 细胞的功能测试和富集,从而为快速开发基于 CAR 的新型疗法提供了有用的研究工具。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Molecular Therapy-Methods & Clinical Development
Biochemistry, Genetics and Molecular Biology-Molecular Biology
CiteScore
9.90
自引率
4.30%
发文量
163
审稿时长
12 weeks
期刊介绍:
The aim of Molecular Therapy—Methods & Clinical Development is to build upon the success of Molecular Therapy in publishing important peer-reviewed methods and procedures, as well as translational advances in the broad array of fields under the molecular therapy umbrella.
Topics of particular interest within the journal''s scope include:
Gene vector engineering and production,
Methods for targeted genome editing and engineering,
Methods and technology development for cell reprogramming and directed differentiation of pluripotent cells,
Methods for gene and cell vector delivery,
Development of biomaterials and nanoparticles for applications in gene and cell therapy and regenerative medicine,
Analysis of gene and cell vector biodistribution and tracking,
Pharmacology/toxicology studies of new and next-generation vectors,
Methods for cell isolation, engineering, culture, expansion, and transplantation,
Cell processing, storage, and banking for therapeutic application,
Preclinical and QC/QA assay development,
Translational and clinical scale-up and Good Manufacturing procedures and process development,
Clinical protocol development,
Computational and bioinformatic methods for analysis, modeling, or visualization of biological data,
Negotiating the regulatory approval process and obtaining such approval for clinical trials.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


