PTPRT loss enhances anti–PD-1 therapy efficacy by regulation of STING pathway in non–small cell lung cancer
IF 15.8
1区 医学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
With the revolutionary progress of immune checkpoint inhibitors (ICIs) in non–small cell lung cancer, identifying patients with cancer who would benefit from ICIs has become critical and urgent. Here, we report protein tyrosine phosphatase receptor type T (PTPRT) loss as a precise and convenient predictive marker independent of PD-L1 expression for anti–PD-1/PD-L1 axis therapy. Anti–PD-1/PD-L1 axis treatment markedly increased progression-free survival in patients with PTPRT-deficient tumors. PTPRT-deficient tumors displayed cumulative DNA damage, increased cytosolic DNA release, and higher tumor mutation burden. Moreover, the tyrosine residue 240 of STING was identified as a direct substrate of PTPRT. PTPRT loss elevated phosphorylation of STING at Y240 and thus inhibited its proteasome-mediated degradation. PTPRT-deficient tumors released more IFN-β, CCL5, and CXCL10 by activation of STING pathway and increased immune cell infiltration, especially of CD8 T cells and natural killer cells, ultimately enhancing the efficacy of anti–PD-1 therapy in multiple subcutaneous and orthotopic tumor mouse models. The response of PTPRT-deficient tumors to anti–PD-1 therapy depends on the tumor-intrinsic STING pathway. In summary, our findings reveal the mechanism of how PTPRT-deficient tumors become sensitive to anti–PD-1 therapy and highlight the biological function of PTPRT in innate immunity. Considering the prevalence of PTPRT mutations and negative expression, this study has great value for patient stratification and clinical decision-making.
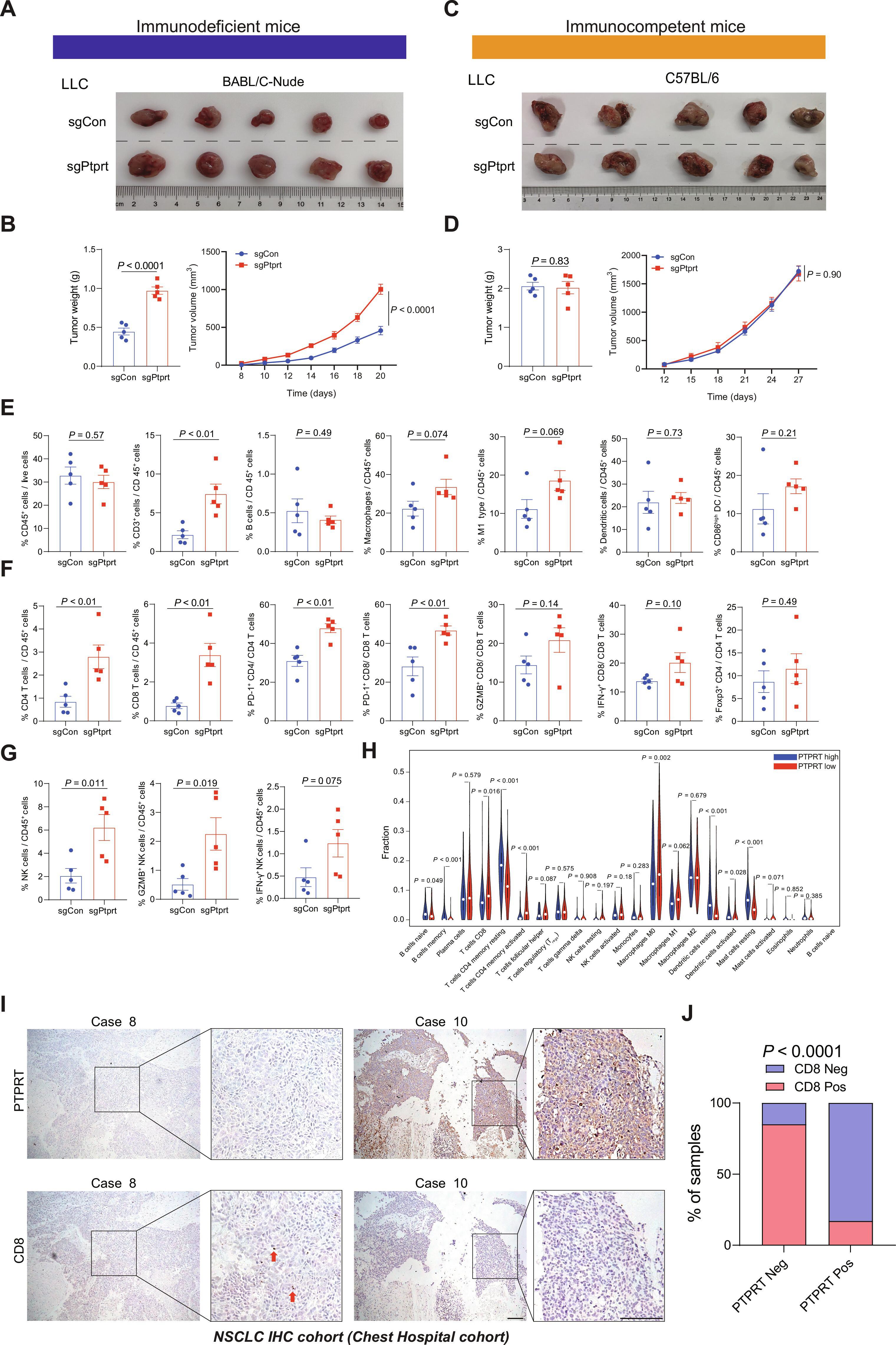
PTPRT缺失可通过调节STING通路增强非小细胞肺癌抗PD-1疗法的疗效。
随着免疫检查点抑制剂(ICIs)在非小细胞肺癌领域取得革命性进展,确定哪些癌症患者可从 ICIs 中获益已变得至关重要且刻不容缓。在此,我们报告了蛋白酪氨酸磷酸酶受体T型(PTPRT)缺失作为一种独立于PD-L1表达的精确、便捷的预测指标,可用于抗PD-1/PD-L1轴治疗。抗PD-1/PD-L1轴治疗显著提高了PTPRT缺失肿瘤患者的无进展生存期。PTPRT缺陷型肿瘤显示出累积性DNA损伤、细胞膜DNA释放增加以及更高的肿瘤突变负荷。此外,STING 的酪氨酸残基 240 被确定为 PTPRT 的直接底物。PTPRT的缺失会增加STING在Y240处的磷酸化,从而抑制蛋白酶体介导的降解。PTPRT 缺失的肿瘤通过激活 STING 通路释放出更多的 IFN-β、CCL5 和 CXCL10,并增加了免疫细胞的浸润,尤其是 CD8 T 细胞和自然杀伤细胞,最终提高了抗 PD-1 治疗在多种皮下和正位肿瘤小鼠模型中的疗效。PTPRT缺陷肿瘤对抗PD-1疗法的反应取决于肿瘤内在STING通路。总之,我们的研究结果揭示了PTPRT缺陷肿瘤如何对抗PD-1治疗变得敏感的机制,并强调了PTPRT在先天免疫中的生物学功能。考虑到PTPRT突变和阴性表达的普遍性,本研究对患者分层和临床决策具有重要价值。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Science Translational Medicine
CELL BIOLOGY-MEDICINE, RESEARCH & EXPERIMENTAL
CiteScore
26.70
自引率
1.20%
发文量
309
审稿时长
1.7 months
期刊介绍:
Science Translational Medicine is an online journal that focuses on publishing research at the intersection of science, engineering, and medicine. The goal of the journal is to promote human health by providing a platform for researchers from various disciplines to communicate their latest advancements in biomedical, translational, and clinical research.
The journal aims to address the slow translation of scientific knowledge into effective treatments and health measures. It publishes articles that fill the knowledge gaps between preclinical research and medical applications, with a focus on accelerating the translation of knowledge into new ways of preventing, diagnosing, and treating human diseases.
The scope of Science Translational Medicine includes various areas such as cardiovascular disease, immunology/vaccines, metabolism/diabetes/obesity, neuroscience/neurology/psychiatry, cancer, infectious diseases, policy, behavior, bioengineering, chemical genomics/drug discovery, imaging, applied physical sciences, medical nanotechnology, drug delivery, biomarkers, gene therapy/regenerative medicine, toxicology and pharmacokinetics, data mining, cell culture, animal and human studies, medical informatics, and other interdisciplinary approaches to medicine.
The target audience of the journal includes researchers and management in academia, government, and the biotechnology and pharmaceutical industries. It is also relevant to physician scientists, regulators, policy makers, investors, business developers, and funding agencies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


