Salinomycin, a potent inhibitor of XOD and URAT1, ameliorates hyperuricemic nephropathy by activating NRF2, modulating the gut microbiota, and promoting SCFA production
Abstract
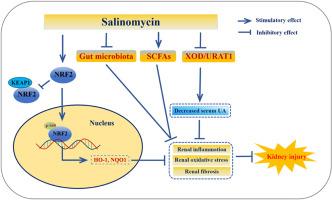
Long-term hyperuricemia can induce kidney damage, clinically referred to as hyperuricemic nephropathy (HN), which is characterized by renal fibrosis, inflammation, and oxidative stress. However, currently used uric acid-lowering drugs are not capable of protecting the kidneys from damage. Therefore, uric acid-lowering drugs that can also protect the kidneys are urgently needed. In this study, we first discovered that salinomycin, an antibiotic, can regulate uric acid homeostasis and ameliorate kidney damage in mice with HN. Mechanistically, salinomycin inhibited serum and hepatic xanthine oxidase (XOD) activities and downregulated renal urate transporter 1 (URAT1) expression and transport activity, thus exerting uric acid-lowering effects in mice with HN. Furthermore, we found that salinomycin promoted p-NRF2 Ser40 expression, resulting in increased nuclear translocation of NRF2 and activation of NRF2. More importantly, salinomycin affected the gut microbiota and promoted the generation of short-chain fatty acids (SCFAs) in mice with HN. In conclusion, our results revealed that salinomycin maintains uric acid homeostasis and alleviates kidney injury in mice with HN by multiple mechanisms, suggesting that salinomycin might be a desirable candidate for HN treatment in the clinic.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


