Clinical utility of BRCA and ATM mutation status in circulating tumour DNA for treatment selection in advanced pancreatic cancer
IF 6.4
1区 医学
Q1 ONCOLOGY
引用次数: 0
Abstract
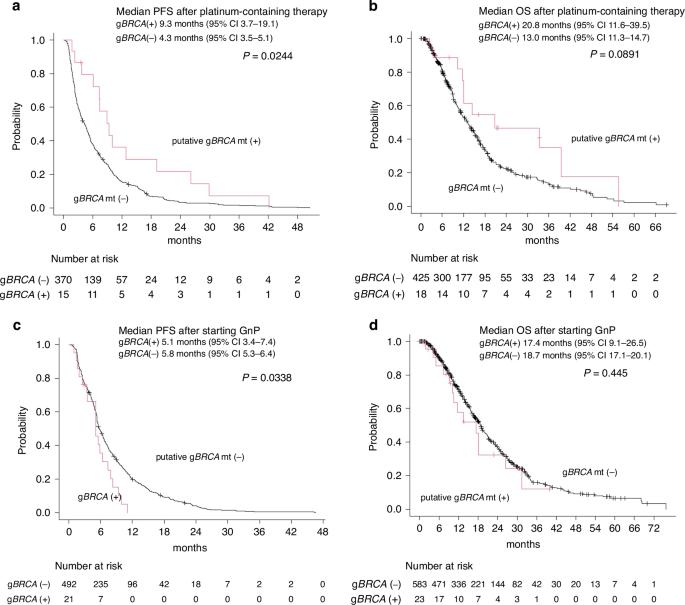
Identification of homologous recombination deficiency (HRD) remains a challenge in advanced pancreatic cancer (APC). We investigated the utility of circulating tumour DNA (ctDNA) profiling in the assessment of BRCA1/2 and ATM mutation status and treatment selection in APC. We analysed clinical and ctDNA data of 702 patients with APC enroled in GOZILA, a ctDNA profiling study using Guardant360. Inactivating BRCA1/2 and ATM mutations were detected in 4.8% (putative germline, 3.7%) and 4.4% (putative germline, 0.9%) of patients, respectively. Objective response (63.2% vs. 16.2%) and PFS (HR 0.55, 95% CI 0.32–0.93) on platinum-containing chemotherapy were significantly better in patients with putative germline BRCA1/2 (gBRCA) mutation than those without. In contrast, putative gBRCA mutation had no impact on the efficacy of gemcitabine plus nab-paclitaxel. In 2 patients treated with platinum-containing therapy, putative BRCA2 reversion mutations were detected. Three of seven patients with somatic BRCA mutations responded to platinum-containing therapy, while only one of four with putative germline ATM mutations did. One-third of somatic ATM mutations were in genomic loci associated with clonal haematopoiesis. Comprehensive ctDNA profiling provides clinically relevant information regarding HRD status. It can be a practical, convenient option for HRD screening in APC.
循环肿瘤 DNA 中 BRCA 和 ATM 突变状态对晚期胰腺癌治疗选择的临床实用性。
背景:在晚期胰腺癌(APC)中,同源重组缺陷(HRD)的鉴定仍是一项挑战。我们研究了循环肿瘤DNA(ctDNA)分析在评估BRCA1/2和ATM突变状态以及胰腺癌治疗选择中的作用:我们分析了702名加入GOZILA的APC患者的临床和ctDNA数据,GOZILA是一项使用Guardant360进行的ctDNA分析研究:结果:4.8%的患者(推定种系,3.7%)和4.4%的患者(推定种系,0.9%)分别检测到灭活BRCA1/2和ATM突变。有推定种系 BRCA1/2 (gBRCA) 基因突变的患者对含铂化疗的客观反应(63.2% 对 16.2%)和 PFS(HR 0.55,95% CI 0.32-0.93)明显优于无基因突变的患者。相比之下,推定的 gBRCA 基因突变对吉西他滨加纳布紫杉醇的疗效没有影响。在接受含铂治疗的两名患者中,检测到了假定的 BRCA2 逆转突变。七名体细胞 BRCA 基因突变的患者中有三人对含铂疗法产生了反应,而四名推定种系 ATM 基因突变的患者中只有一人产生了反应。三分之一的体细胞ATM突变位于与克隆性造血相关的基因组位点:结论:全面的ctDNA分析可提供有关HRD状态的临床相关信息。结论:全面的ctDNA分析可提供有关HRD状态的临床相关信息,是APC中HRD筛查的一种实用、方便的选择。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

British Journal of Cancer
医学-肿瘤学
CiteScore
15.10
自引率
1.10%
发文量
383
审稿时长
6 months
期刊介绍:
The British Journal of Cancer is one of the most-cited general cancer journals, publishing significant advances in translational and clinical cancer research.It also publishes high-quality reviews and thought-provoking comment on all aspects of cancer prevention,diagnosis and treatment.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


