Comparison of Rocuronium and Succinylcholine for First-Attempt Intubation Success in the Emergency Department
Abstract
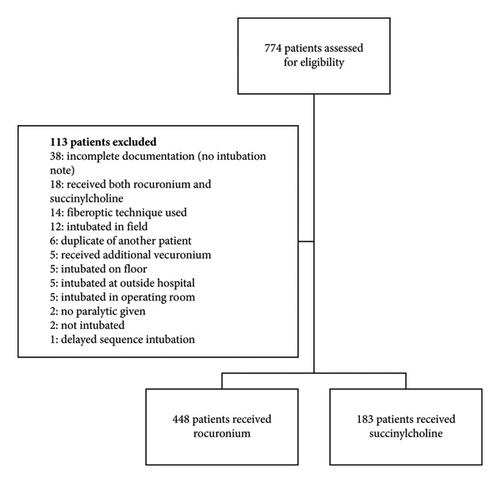
Background. Succinylcholine and rocuronium are the predominant neuromuscular blocking agents (NMBAs) used for rapid sequence intubation (RSI) in the emergency department (ED). Prior studies have found reduced first-attempt intubation success (FAIS) with rocuronium compared to succinylcholine. Recent large registry data have shown no difference in intubating conditions or FAIS. Objectives. The objective of this study was to compare FAIS rates for rocuronium and succinylcholine when used for RSI in a high-acuity academic ED. Methods. This was a single-center retrospective study. Patients were included if they received either succinylcholine or rocuronium for RSI in the ED from January 2016 to August 2020. The primary endpoint was FAIS. Subgroup analyses were performed evaluating the impact of weight-based dosing on FAIS for each agent, and multivariate analysis was conducted to evaluate the impact of baseline characteristics on the primary outcome. Results. There were 448 patients who received rocuronium and 183 patients who received succinylcholine. No difference was observed in unadjusted FAIS between patients receiving rocuronium (median weight-based dose: 1.22 mg/kg) or succinylcholine (median weight-based dose: 1.43 mg/kg) (361 (80.6%) vs. 150 (82.0%), p = 0.69). There were no differences in FAIS between the weight-based dose categories for rocuronium and for succinylcholine. Conclusions. These findings were consistent with those from recent studies indicating no difference in FAIS between rocuronium and succinylcholine, although the median dose of rocuronium used in this study was higher than traditionally recommended. Larger prospective studies are warranted to further evaluate the effect of weight-based paralytic dosing on FAIS.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


