Engineering Central Substitutions in Heptamethine Dyes for Improved Fluorophore Performance
引用次数: 0
Abstract
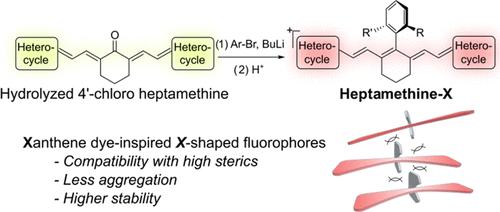
As a major family of red-shifted fluorophores that operate beyond visible light, polymethine dyes are pivotal in light-based biological techniques. However, methods for tuning this kind of fluorophores by structural modification remain restricted to bottom-up synthesis and modification using coupling or nucleophilic substitutions. In this study, we introduce a two-step, late-stage functionalization process for heptamethine dyes. This process enables the substitution of the central chlorine atom in the commonly used 4′-chloro heptamethine scaffold with various aryl groups using aryllithium reagents. This method borrows the building block and designs from the xanthene dye community and offers a mild and convenient way for the diversification of heptamethine fluorophores. Notably, this efficient conversion allows for the synthesis of heptamethine-X, the heptamethine scaffold with two ortho-substituents on the 4′-aryl modification, which brings enhanced stability and reduced aggregation to the fluorophore. We showcase the utility of this method by a facile synthesis of a fluorogenic, membrane-localizing fluorophore that outperforms its commercial counterparts with a significantly higher brightness and contrast. Overall, this method establishes the synthetic similarities between polymethine and xanthene fluorophores and provides a versatile and feasible toolbox for future optimizing heptamethine fluorophores for their biological applications.
在七甲基染料中进行中心取代工程以提高发荧光体的性能
作为在可见光以外工作的红移荧光团的一个主要家族,聚甲基染料在基于光的生物技术中举足轻重。然而,通过结构修饰来调整这类荧光团的方法仍局限于自下而上的合成和利用偶联或亲核取代进行修饰。在本研究中,我们介绍了七甲基染料的两步后期功能化工艺。该工艺可使用芳基锂试剂将常用的 4′-氯七甲基支架中的中心氯原子替换为各种芳基。这种方法借鉴了呫吨染料的结构单元和设计,为七甲基荧光团的多样化提供了一种温和、便捷的方法。值得注意的是,这种高效的转化方法可以合成庚氨酸-X,即在 4′-芳基修饰上带有两个正交取代基的庚氨酸支架,从而提高了荧光团的稳定性并减少了聚集。我们展示了这种方法的实用性,它简便地合成了一种含氟的膜定位荧光团,其亮度和对比度明显高于商业同类荧光团。总之,这种方法确定了聚甲基和氧杂蒽荧光团之间的合成相似性,并为今后优化七甲基荧光团的生物应用提供了一个多功能、可行的工具箱。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


