Ketogenic diet reshapes cancer metabolism through lysine β-hydroxybutyrylation
IF 20.8
1区 医学
Q1 ENDOCRINOLOGY & METABOLISM
引用次数: 0
Abstract
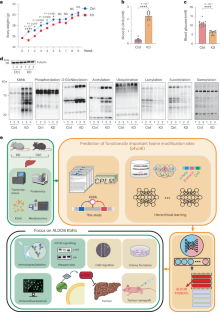
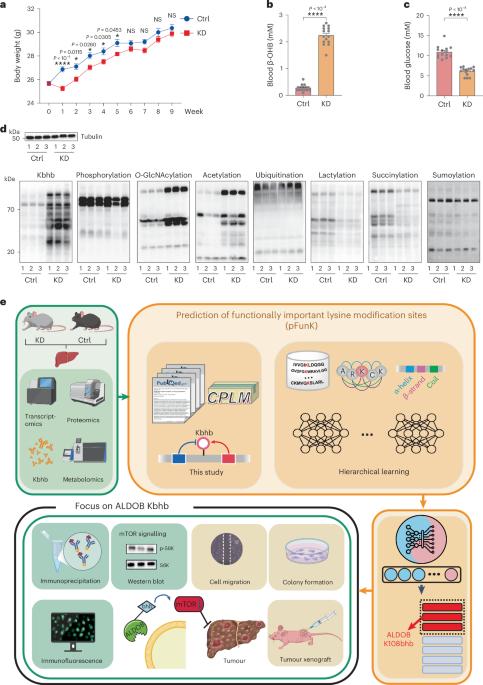
Lysine β-hydroxybutyrylation (Kbhb) is a post-translational modification induced by the ketogenic diet (KD), a diet showing therapeutic effects on multiple human diseases. Little is known how cellular processes are regulated by Kbhb. Here we show that protein Kbhb is strongly affected by the KD through a multi-omics analysis of mouse livers. Using a small training dataset with known functions, we developed a bioinformatics method for the prediction of functionally important lysine modification sites (pFunK), which revealed functionally relevant Kbhb sites on various proteins, including aldolase B (ALDOB) Lys108. KD consumption or β-hydroxybutyrate supplementation in hepatocellular carcinoma cells increases ALDOB Lys108bhb and inhibits the enzymatic activity of ALDOB. A Kbhb-mimicking mutation (p.Lys108Gln) attenuates ALDOB activity and its binding to substrate fructose-1,6-bisphosphate, inhibits mammalian target of rapamycin signalling and glycolysis, and markedly suppresses cancer cell proliferation. Our study reveals a critical role of Kbhb in regulating cancer cell metabolism and provides a generally applicable algorithm for predicting functionally important lysine modification sites. Multi-omics analysis of mouse livers shows that protein β-hydroxybutyrylation (Kbhb) is strongly affected by the ketogenic diet. Combining bioinformatics with experimental validation, a role of Kbhb sites in modulating mammalian target of rapamycin signalling and cancer cell metabolism is identified.
生酮饮食通过赖氨酸β-羟基丁酰化重塑癌症代谢
赖氨酸β-羟基丁酰化(Kbhb)是生酮饮食(KD)诱导的一种翻译后修饰,这种饮食对多种人类疾病有治疗作用。人们对 Kbhb 如何调控细胞过程知之甚少。在这里,我们通过对小鼠肝脏的多组学分析表明,蛋白质 Kbhb 受到生酮饮食的强烈影响。利用已知功能的小型训练数据集,我们开发了一种预测重要功能赖氨酸修饰位点(pFunK)的生物信息学方法,该方法揭示了各种蛋白质上与功能相关的 Kbhb 位点,包括醛缩酶 B (ALDOB) Lys108。肝癌细胞摄入 KD 或补充β-羟丁酸会增加 ALDOB Lys108bhb,并抑制 ALDOB 的酶活性。Kbhb模拟突变(p.Lys108Gln)会减弱ALDOB的活性及其与底物1,6-二磷酸果糖的结合,抑制雷帕霉素哺乳动物靶标信号传导和糖酵解,并显著抑制癌细胞增殖。我们的研究揭示了 Kbhb 在调节癌细胞新陈代谢中的关键作用,并为预测重要的赖氨酸修饰位点提供了一种普遍适用的算法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
Nature metabolism
ENDOCRINOLOGY & METABOLISM-
CiteScore
27.50
自引率
2.40%
发文量
170
期刊介绍:
Nature Metabolism is a peer-reviewed scientific journal that covers a broad range of topics in metabolism research. It aims to advance the understanding of metabolic and homeostatic processes at a cellular and physiological level. The journal publishes research from various fields, including fundamental cell biology, basic biomedical and translational research, and integrative physiology. It focuses on how cellular metabolism affects cellular function, the physiology and homeostasis of organs and tissues, and the regulation of organismal energy homeostasis. It also investigates the molecular pathophysiology of metabolic diseases such as diabetes and obesity, as well as their treatment. Nature Metabolism follows the standards of other Nature-branded journals, with a dedicated team of professional editors, rigorous peer-review process, high standards of copy-editing and production, swift publication, and editorial independence. The journal has a high impact factor, has a certain influence in the international area, and is deeply concerned and cited by the majority of scholars.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


