A noncanonical E3 ubiquitin ligase RNF41-mediated MYO1C stability promotes prostate cancer metastasis by inducing actin remodeling
IF 6.9
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
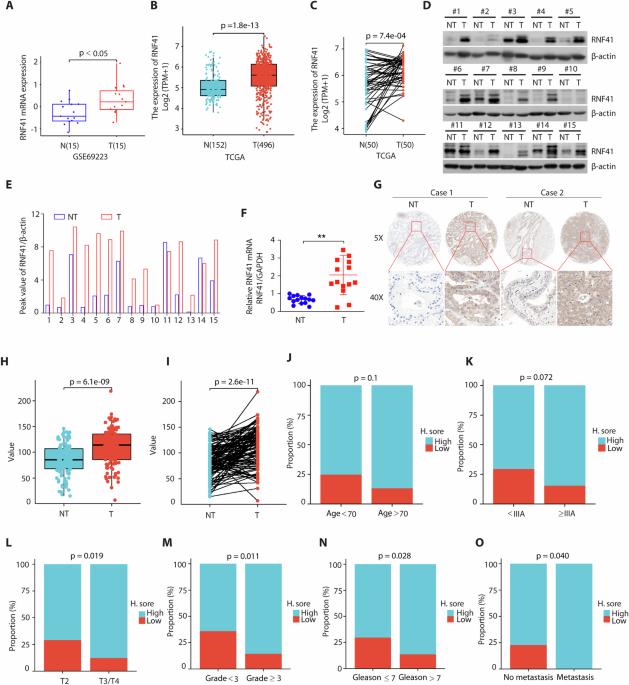
Prostate cancer bone metastasis is a predominant cause of death for prostate cancer (PCa) patients. However, the underlying mechanisms are poorly understood. Here, we report that high levels of RNF41 are associated with metastatic human prostate cancer. RNF41 silencing inhibits prostate cancer cell growth, cell migration and invasion in vitro and in vivo. Mechanistically, we identify that RNF41 induces K27- and K63-linked noncanonical polyubiquitination of MYO1C to enhance its stability and induce actin remodeling, which promotes PCa bone metastasis. RNF41 was significantly upregulated in metastatic prostate cancer tissues and positively associated with MYO1C expression. Furthermore, we show in intraarterial injected-bone metastasis xenograft model that targeting MYO1C stability by inhibition of RNF41 markedly suppressed PCa bone metastasis. Collectively, our findings identify RNF41 is an important regulator of prostate cancer cell growth and metastasis and targeting RNF41/MYO1C could be a valuable strategy to ameliorate prostate cancer progression and metastasis.
非规范E3泛素连接酶RNF41介导的MYO1C稳定性通过诱导肌动蛋白重塑促进前列腺癌转移。
前列腺癌骨转移是前列腺癌(PCa)患者的主要死因。然而,人们对其潜在机制知之甚少。在此,我们报告了高水平的 RNF41 与转移性人类前列腺癌有关。沉默 RNF41 可抑制前列腺癌细胞在体外和体内的生长、迁移和侵袭。从机理上讲,我们发现 RNF41 会诱导 MYO1C 的 K27- 和 K63 链接的非规范多泛素化,从而增强其稳定性并诱导肌动蛋白重塑,从而促进 PCa 骨转移。RNF41 在转移性前列腺癌组织中明显上调,并与 MYO1C 的表达呈正相关。此外,我们在动脉内注射骨转移异种移植模型中发现,通过抑制 RNF41 来靶向 MYO1C 的稳定性能明显抑制 PCa 骨转移。总之,我们的研究结果表明,RNF41是前列腺癌细胞生长和转移的重要调控因子,靶向RNF41/MYO1C可能是改善前列腺癌进展和转移的一种有价值的策略。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Oncogene
医学-生化与分子生物学
CiteScore
15.30
自引率
1.20%
发文量
404
审稿时长
1 months
期刊介绍:
Oncogene is dedicated to advancing our understanding of cancer processes through the publication of exceptional research. The journal seeks to disseminate work that challenges conventional theories and contributes to establishing new paradigms in the etio-pathogenesis, diagnosis, treatment, or prevention of cancers. Emphasis is placed on research shedding light on processes driving metastatic spread and providing crucial insights into cancer biology beyond existing knowledge.
Areas covered include the cellular and molecular biology of cancer, resistance to cancer therapies, and the development of improved approaches to enhance survival. Oncogene spans the spectrum of cancer biology, from fundamental and theoretical work to translational, applied, and clinical research, including early and late Phase clinical trials, particularly those with biologic and translational endpoints.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


