Intermolecular Forces and the Languages of Chemistry
IF 2.5
3区 教育学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
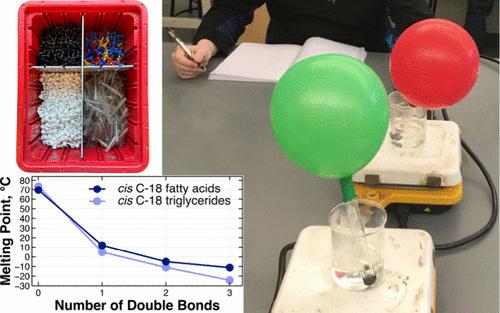
The concept of intermolecular forces is challenging because students often revert to intramolecular explanations when discussing macroscopic properties. This set of activities from our general chemistry course aimed to use the different languages of chemistry to help build a more solid, multilevel understanding of intermolecular forces. Through the symbolic language of graphing, students were first asked to explore the relationship among melting point, molecular weight, and structure. Macroscopic language was used during a lab activity where students hydrogenated common kitchen oils and saw a change from liquid to solid. In another activity, students employed atomic/molecular/particulate language in building models and thinking about the interactions between molecules. They were asked to make use of all three languages in examining the intermolecular forces relevant to the melting points of fatty acids and triglycerides.
分子间作用力与化学语言
分子间作用力的概念具有挑战性,因为学生在讨论宏观性质时往往会回到分子内解释。我们普通化学课程的这组活动旨在使用不同的化学语言,帮助学生建立对分子间作用力更扎实、多层次的理解。首先,通过图形的符号语言,要求学生探索熔点、分子量和结构之间的关系。在一次实验活动中,学生将厨房常用的油氢化,看到了从液态到固态的变化。在另一项活动中,学生们使用原子/分子/微粒语言建立模型并思考分子间的相互作用。在研究与脂肪酸和甘油三酯熔点有关的分子间作用力时,他们被要求使用这三种语言。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Journal of Chemical Education
化学-化学综合
CiteScore
5.60
自引率
50.00%
发文量
465
审稿时长
6.5 months
期刊介绍:
The Journal of Chemical Education is the official journal of the Division of Chemical Education of the American Chemical Society, co-published with the American Chemical Society Publications Division. Launched in 1924, the Journal of Chemical Education is the world’s premier chemical education journal. The Journal publishes peer-reviewed articles and related information as a resource to those in the field of chemical education and to those institutions that serve them. JCE typically addresses chemical content, activities, laboratory experiments, instructional methods, and pedagogies. The Journal serves as a means of communication among people across the world who are interested in the teaching and learning of chemistry. This includes instructors of chemistry from middle school through graduate school, professional staff who support these teaching activities, as well as some scientists in commerce, industry, and government.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


