DFT Study on Retigerane-Type Sesterterpenoid Biosynthesis: Initial Conformation of GFPP Regulates Biosynthetic Pathway, Ring-Construction Order and Stereochemistry
引用次数: 0
Abstract
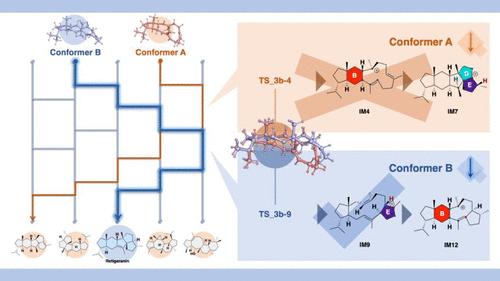
Retigerane-type sesterterpenoids, which feature a unique 5/6/5/5/5 fused pentacyclic structure with an angular-type triquinane moiety, are biosynthesized via successive carbocation-mediated reactions triggered by terpene cyclases. However, the precise biosynthetic pathways/mechanisms, wherein steric inversion of the carbon skeleton occurs at least once, remain elusive. Two plausible reaction pathways have been proposed, which differ in the order of ring cyclization: A → B/C → D/E-ring(s) (Route 1) and A → E → B → C/D-ring(s) (Route 2). Since the reaction intermediates of these complicated domino-type reaction sequences are experimentally inaccessible, we employed comprehensive density functional theory (DFT) calculations to evaluate these routes. The results indicate that retigeranin biosynthesis proceeds via Route 2 involving a multistep carbocation cascade, in which the order of ring cyclization (A → E → B → C/D) is the key to constructing the angular 5/5/5 triquinane structure with the correct stereochemistry at C3. The result also suggests that slight differences in the initial conformation have a significant effect on the order of cyclization and steric inversion. The computed pathway/mechanism also provides a rational basis for the formation of various related terpenes/terpenoids.
芸苔素类酯萜生物合成的 DFT 研究:GFPP 的初始构象调节生物合成途径、环结构顺序和立体化学
芸苔素类酯萜类化合物具有独特的 5/6/5/5/5 融合五环结构和角型三喹啉分子,通过萜烯环化酶引发的连续碳代反应进行生物合成。然而,碳骨架至少发生一次立体反转的精确生物合成途径/机制仍然难以确定。人们提出了两种可信的反应途径,它们在环化顺序上有所不同:A → B/C → D/E 环(路线 1)和 A → E → B → C/D 环(路线 2)。由于这些复杂的多米诺型反应序列的反应中间体无法通过实验获得,我们采用了全面的密度泛函理论(DFT)计算来评估这些路线。结果表明,瑞格素的生物合成是通过路线 2 进行的,其中涉及一个多步骤的碳位级联反应,环的环化顺序(A → E → B → C/D)是构建具有正确 C3 立体化学结构的角 5/5/5 三喹啉结构的关键。该结果还表明,初始构象的细微差别对环化和立体反转的顺序有显著影响。计算出的途径/机制还为各种相关萜类/三萜类化合物的形成提供了合理的依据。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


