YAP mediates HIV-related liver fibrosis
IF 9.5
1区 医学
Q1 GASTROENTEROLOGY & HEPATOLOGY
引用次数: 0
Abstract
Background & Aims
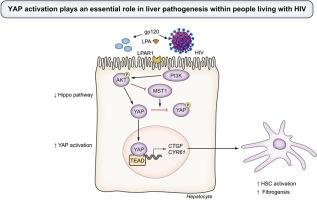
HIV accelerates liver fibrosis attributable to multiple etiologies, including HCV, HBV, and steatotic liver disease. Evidence also suggests that HIV infection itself is associated with liver fibrogenesis. Recent studies have implicated Yes-associated protein 1 (YAP1) and the upstream lysophosphatidic acid (LPA)/PI3K/AKT pathway as critical regulators of hepatic fibrogenesis, and suggest a connection to HIV-related liver fibrosis. However, the relationship between YAP/PI3K/AKT pathway activation and HIV-related liver fibrosis remains uncertain.
Methods
qPCR, western blot, immunofluorescence, and ELISA (replicates n ≥3) were performed in an unbiased humanized mouse model (NRG-hu HSC mice, n = 6), the precision cut liver slice ex vivo model, and both traditional in vitro models as well as a 3D spheroid system.
Results
YAP target gene mRNA and protein levels (ANKRD, CTGF, CYR61) were upregulated across all models exposed to HIV. Humanized mice infected with HIV had significant increases in the percentage of YAP-positive nuclei (2.2-fold) and the percentage area of Sirius Red collagen staining (3.3-fold) compared to control mice. Serum concentrations of LPA were increased 5.8-fold in people living with HIV compared to healthy controls. Modulation of LPAR1, PI3K, and AKT by either inhibitors or small-interfering RNAs abrogated the fibrotic effects of HIV exposure and downregulated YAP target genes within cultured liver cells.
Conclusions
The LPAR/PI3K/AKT axis is vital for the activation of YAP and hepatic fibrogenesis due to HIV infection. This novel mechanistic insight suggests new pharmacologic targets for treatment of liver fibrosis in people living with HIV.
Impact and implications:
There are currently no FDA-approved treatments for cirrhosis, while liver disease is the second leading cause of mortality among people living with HIV after AIDS. Increased lysophosphatidic acid concentrations and AKT activation after HIV infection found in recent work suggest that the Hippo pathway may be a key regulator of HIV-related fibrogenesis. By linking lysophosphatidic acid signaling, YAP activation, and HIV-related fibrogenesis, this mechanism presents a target for future research into therapeutic interventions for not only HIV but also other liver diseases, e.g. metabolic dysfunction- or alcohol-associated liver disease.
YAP 介导与艾滋病毒相关的肝纤维化
背景& 目的HIV会加速多种病因引起的肝纤维化,包括HCV、HBV和脂肪肝。还有证据表明,HIV 感染本身也与肝纤维化有关。最近的研究表明,Yes 相关蛋白 1(YAP1)和上游溶血磷脂酸(LPA)/PI3K/AKT 通路是肝纤维化的关键调节因子,并与 HIV 相关的肝纤维化有关。然而,YAP/PI3K/AKT 通路激活与 HIV 相关肝纤维化之间的关系仍不确定。方法在无偏见的人源化小鼠模型(NRG-hu HSC 小鼠,n = 6)、精确切割肝切片体外模型、传统体外模型以及三维球状系统中进行了 qPCR、Western 印迹、免疫荧光和 ELISA(重复 n ≥3)。结果YAP靶基因mRNA和蛋白水平(ANKRD、CTGF、CYR61)在所有暴露于HIV的模型中均上调。与对照组小鼠相比,感染艾滋病毒的人源化小鼠的 YAP 阳性细胞核百分比(2.2 倍)和天狼星红胶原染色面积百分比(3.3 倍)显著增加。与健康对照组相比,HIV 感染者血清中的 LPA 浓度增加了 5.8 倍。通过抑制剂或小干扰 RNA 对 LPAR1、PI3K 和 AKT 进行调节,可减轻 HIV 暴露对肝纤维化的影响,并下调培养肝细胞中的 YAP 靶基因。这一新颖的机理见解为治疗艾滋病病毒感染者的肝纤维化提出了新的药物靶点。影响和意义:目前,美国食品及药物管理局还没有批准治疗肝硬化的药物,而肝病是艾滋病病毒感染者继艾滋病之后的第二大死亡原因。近期研究发现,HIV感染后溶血磷脂酸浓度升高和AKT活化表明,Hippo通路可能是HIV相关纤维化的关键调节因子。通过将溶血磷脂酸信号传导、YAP 激活和 HIV 相关纤维形成联系起来,这一机制为未来研究治疗干预措施提供了一个目标,不仅可以治疗 HIV,还可以治疗其他肝病,如代谢功能障碍或酒精相关肝病。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

JHEP Reports
GASTROENTEROLOGY & HEPATOLOGY-
CiteScore
12.40
自引率
2.40%
发文量
161
审稿时长
36 days
期刊介绍:
JHEP Reports is an open access journal that is affiliated with the European Association for the Study of the Liver (EASL). It serves as a companion journal to the highly respected Journal of Hepatology.
The primary objective of JHEP Reports is to publish original papers and reviews that contribute to the advancement of knowledge in the field of liver diseases. The journal covers a wide range of topics, including basic, translational, and clinical research. It also focuses on global issues in hepatology, with particular emphasis on areas such as clinical trials, novel diagnostics, precision medicine and therapeutics, cancer research, cellular and molecular studies, artificial intelligence, microbiome research, epidemiology, and cutting-edge technologies.
In summary, JHEP Reports is dedicated to promoting scientific discoveries and innovations in liver diseases through the publication of high-quality research papers and reviews covering various aspects of hepatology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


