Design and Efficient Synthesis of New 4-Amino Substituted 2-(4-bromobenzyl)-5,6,7,8-tetrahydrobenzo[4,5]thieno[2,3-d]pyrimidines of Anticancer Interest and their In Silico Study
引用次数: 0
Abstract
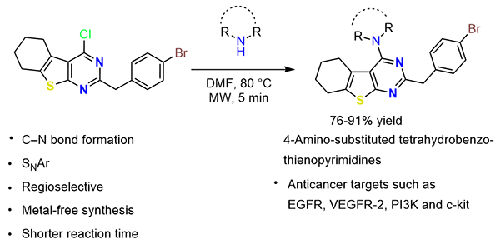
Thienopyrimidines are one of the emerging classes among fused pyrimidines owing to their broad spectrum of pharmacological properties, including antimicrobial, anti-inflammatory, antimalarial, anticancer, etc. The anticancer activity of these compounds is mechanistically proven via the inhibition of validated drug targets such as EGFR, VEGFR-2, PI3K, and c-kit. In this research article, we designed and attempted synthesis of new 4-amino substituted 2-(4-bromobenzyl)-5,6,7,8-tetrahydrobenzo[4,5]thieno[2,3-d]pyrimidines to explore their anticancer potential further. These heterocycles were designed based on pharmacophoric features of the core heterocycle, varying its C-4 substitution with a variety of amines and considering cancer protein-ligand interactions aiming to get potent lead molecules. The target compound-protein interaction complexes were analyzed, and lead compounds were identified based on their better binding affinity in molecular docking studies.
具有抗癌意义的新型 4-氨基取代 2-(4-溴苄基)-5,6,7,8-四氢苯并[4,5]噻吩并[2,3-d]嘧啶的设计与高效合成及其硅学研究
噻吩嘧啶类化合物具有抗菌、消炎、抗疟、抗癌等多种药理特性,是新兴的融合嘧啶类化合物之一。通过抑制 EGFR、VEGFR-2、PI3K 和 c-kit 等有效药物靶点,这些化合物的抗癌活性得到了机理上的证实。在这篇研究文章中,我们设计并尝试合成了新的 4-氨基取代的 2-(4-溴苄基)-5,6,7,8-四氢苯并[4,5]噻吩并[2,3-d]嘧啶,以进一步探索其抗癌潜力。这些杂环的设计基于核心杂环的药效学特征、C-4 与各种胺的不同取代度以及癌症蛋白配体之间的相互作用,旨在获得有效的先导分子。对目标化合物-蛋白质相互作用复合物进行了分析,并根据分子对接研究中更好的结合亲和力确定了先导化合物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


