Hydrogen-Bonding Activation of Gold(I) Chloride Complexes: Enantioselective Synthesis of 3(2H)-Furanones by a Cycloisomerization-Addition Cascade
IF 4.9
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Enantioselective synthesis of 3(2H)-furanones has been achieved using the intermolecular H-bonding activation of gold(I) chloride complexes. A DM-BINAP [(R)-(+)-2,2′-Bis[di(3,5-xylyl)phoshino]-1,1′-binaphthyl] digold(I) dichloride complex in combination with a sulfonyl squaramide (SO2Sq) has been identified as the optimal catalytic system. The process involves a 5-endo-dig oxa-cyclization followed by stereocontrolled addition of indoles. Interestingly, the soft L*Au–Cl activation by H-bonding allowed the recovery of both L*Au–Cl and the activator after chromatographic purification.
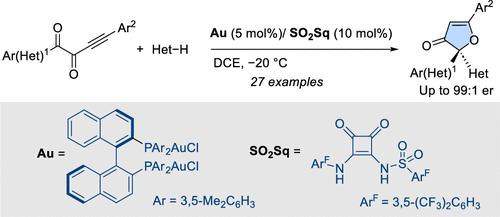
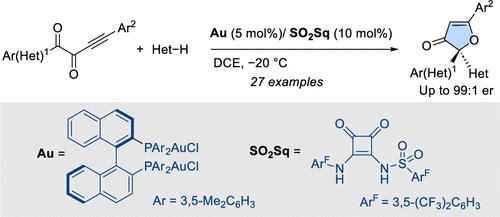
金(I)氯化物配合物的氢键活化:通过环异构化-加成级联法对 3(2H)-呋喃酮进行对映选择性合成。
利用氯化金(I)络合物的分子间 H 键活化实现了 3(2H)-呋喃酮的对映选择性合成。DM-BINAP [(R)-(+)-2,2'-Bis[di(3,5-xylyl)phoshino]-1,1'-binaphthyl] 二氯化金(I)络合物与磺酰方酰胺 (SO2Sq) 组合被确定为最佳催化体系。该过程包括 5-内向二氧环化,然后是吲哚的立体控制加成。有趣的是,通过 H 键的软 L*Au-Cl 活化,可以在色谱纯化后同时回收 L*Au-Cl 和活化剂。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic Letters
化学-有机化学
CiteScore
9.30
自引率
11.50%
发文量
1607
审稿时长
1.5 months
期刊介绍:
Organic Letters invites original reports of fundamental research in all branches of the theory and practice of organic, physical organic, organometallic,medicinal, and bioorganic chemistry. Organic Letters provides rapid disclosure of the key elements of significant studies that are of interest to a large portion of the organic community. In selecting manuscripts for publication, the Editors place emphasis on the originality, quality and wide interest of the work. Authors should provide enough background information to place the new disclosure in context and to justify the rapid publication format. Back-to-back Letters will be considered. Full details should be reserved for an Article, which should appear in due course.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


