Discovery of highly potent phosphodiesterase-1 inhibitors by a combined-structure free energy perturbation approach
IF 14.7
1区 医学
Q1 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Accurate receptor/ligand binding free energy calculations can greatly accelerate drug discovery by identifying highly potent ligands. By simulating the change from one compound structure to another, the relative binding free energy (RBFE) change can be calculated based on the theoretically rigorous free energy perturbation (FEP) method. However, existing FEP-RBFE approaches may face convergence challenges due to difficulties in simulating non-physical intermediate states, which can lead to increased computational costs to obtain the converged results. To fundamentally overcome these issues and accelerate drug discovery, a new combined-structure RBFE (CS-FEP) calculation strategy was proposed, which solved the existing issues by constructing a new alchemical pathway, smoothed the alchemical transformation, increased the phase-space overlap between adjacent states, and thus significantly increased the convergence and accelerated the relative binding free energy calculations. This method was extensively tested in a practical drug discovery effort by targeting phosphodiesterase-1 (PDE1). Starting from a PDE1 inhibitor (compound 9, IC50 = 16.8 μmol/L), the CS-FEP guided hit-to-lead optimizations resulted in a promising lead (11b and its mesylate salt formulation 11b-Mesylate, IC50 = 7.0 nmol/L), with ∼2400-fold improved inhibitory activity. Further experimental studies revealed that the lead showed reasonable metabolic stability and significant anti-fibrotic effects in vivo.

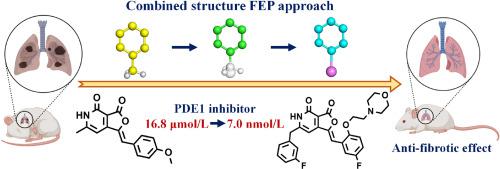
通过联合结构自由能扰动方法发现高效磷酸二酯酶-1 抑制剂
精确的受体/配体结合自由能计算可以通过识别高效力配体来大大加快药物发现的速度。通过模拟从一种化合物结构到另一种化合物结构的变化,可以根据理论上严格的自由能扰动(FEP)方法计算出相对结合自由能(RBFE)的变化。然而,由于难以模拟非物理中间状态,现有的 FEP-RBFE 方法可能面临收敛难题,从而导致获得收敛结果的计算成本增加。为了从根本上克服这些问题,加速药物发现,我们提出了一种新的组合结构 RBFE(CS-FEP)计算策略,通过构建新的炼金术途径、平滑炼金术转化、增加相邻态之间的相空间重叠来解决现有问题,从而显著提高了收敛性并加速了相对结合自由能的计算。这种方法以磷酸二酯酶-1(PDE1)为目标,在实际药物发现工作中进行了广泛测试。从一个 PDE1 抑制剂(化合物,IC = 16.8 μmol/L)开始,CS-FEP 引导的 "命中先导"(hit-to-lead)优化产生了一个很有前途的先导(及其甲磺酸盐制剂 -Mesylate,IC = 7.0 nmol/L),其抑制活性提高了 2400 倍。进一步的实验研究表明,该先导物具有合理的代谢稳定性和显著的抗纤维化作用。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Acta Pharmaceutica Sinica. B
Pharmacology, Toxicology and Pharmaceutics-General Pharmacology, Toxicology and Pharmaceutics
CiteScore
22.40
自引率
5.50%
发文量
1051
审稿时长
19 weeks
期刊介绍:
The Journal of the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association oversees the peer review process for Acta Pharmaceutica Sinica. B (APSB).
Published monthly in English, APSB is dedicated to disseminating significant original research articles, rapid communications, and high-quality reviews that highlight recent advances across various pharmaceutical sciences domains. These encompass pharmacology, pharmaceutics, medicinal chemistry, natural products, pharmacognosy, pharmaceutical analysis, and pharmacokinetics.
A part of the Acta Pharmaceutica Sinica series, established in 1953 and indexed in prominent databases like Chemical Abstracts, Index Medicus, SciFinder Scholar, Biological Abstracts, International Pharmaceutical Abstracts, Cambridge Scientific Abstracts, and Current Bibliography on Science and Technology, APSB is sponsored by the Institute of Materia Medica, Chinese Academy of Medical Sciences, and the Chinese Pharmaceutical Association. Its production and hosting are facilitated by Elsevier B.V. This collaborative effort ensures APSB's commitment to delivering valuable contributions to the pharmaceutical sciences community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


