Mass Spectrometry-Based Protein Footprinting Defines the Binding Pocket of Crotonylated H3K14 in the PHD1 Domain of BAF45D within the BAF Chromatin Remodeling Complex
IF 3.8
Q2 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
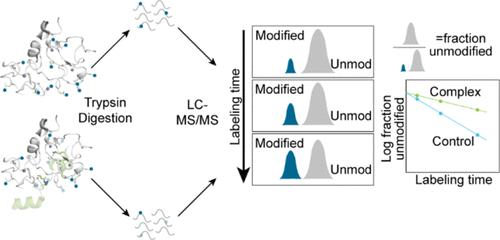
The BRG-/BRM-associated factor (BAF) chromatin remodeling complex is a central actor in transcription. One mechanism by which BAF affects gene expression is via its various histone mark readers, including double plant homeodomains (DPF), located in the BAF45D subunit. DPF domains recognize lysine acetyl and acylations, including crotonylation, localized at promoters and enhancers. Despite a significant degree of conservation between DPF domains, attempts to crystallize BAF45D with a crotonylated histone 3 peptide (H3K14Cr) were unsuccessful. In addition, recent cryoEM and modeled structures failed to define the Req domain of BAF45D, which is responsible for reading lysine modifications. Thus, the precise mechanism of crotonyl group recognition and binding by BAF45D within the BAF complex remains unclear. We turned to protein footprinting mass spectrometry to map the binding interface between H3K14Cr and BAF45D. This technique is able to demarcate protein-binding interfaces by modifying surface-accessible residues and is not limited by protein size or composition. Experiments performed in the isolated DPF domain of BAF45D (BAF45DDPF)-delineated H3K14Cr peptide binding across the PHD1 and PHD2 pockets. We observed markedly similar effects on the BAF45D subunit when assessing H3K14Cr binding in the purified full BAF complex. The ATPase motor, BRM, also displayed H3K14Cr-protected peptides in two separate domains that were subsequently evaluated in direct binding assays. These data confirm the BAF45D–crotonylamide interaction within its obligate complex and are the first to demonstrate H3K14Cr direct binding to BRM.
基于质谱的蛋白质足迹分析确定了 BAF 染色质重塑复合物中 BAF45D 的 PHD1 结构域中克罗顿化 H3K14 的结合位点
BRG-/BRM-相关因子(BAF)染色质重塑复合体是转录过程中的核心角色。BAF 影响基因表达的机制之一是通过其各种组蛋白标记阅读器,包括位于 BAF45D 亚基中的双植物同源结构域(DPF)。DPF 结构域能识别启动子和增强子上的赖氨酸乙酰化和酰化,包括巴豆酰化。尽管 DPF 结构域之间有很大程度的保守性,但试图将 BAF45D 与巴豆酰化组蛋白 3 多肽(H3K14Cr)结晶的尝试并不成功。此外,最近的冷冻电镜和模型结构也未能确定 BAF45D 负责读取赖氨酸修饰的 Req 结构域。因此,BAF45D 在 BAF 复合物中识别巴豆酰基并与之结合的确切机制仍不清楚。我们转而采用蛋白质足迹质谱法来绘制 H3K14Cr 与 BAF45D 之间的结合界面。这种技术能够通过修饰表面可触及的残基来划分蛋白质结合界面,而且不受蛋白质大小或组成的限制。在分离的 BAF45D 的 DPF 结构域(BAF45DDPF)中进行的实验划定了 H3K14Cr 肽与 PHD1 和 PHD2 口袋的结合。在评估纯化的完整 BAF 复合物中的 H3K14Cr 结合情况时,我们观察到 BAF45D 亚基受到了明显类似的影响。ATPase 马达 BRM 也在两个独立的结构域中显示了 H3K14Cr 保护肽,随后在直接结合试验中对其进行了评估。这些数据证实了 BAF45D 与巴豆酰酰胺在其必须复合体中的相互作用,并首次证明了 H3K14Cr 与 BRM 的直接结合。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Bio & Med Chem Au
药物、生物、化学-
CiteScore
4.10
自引率
0.00%
发文量
0
期刊介绍:
ACS Bio & Med Chem Au is a broad scope open access journal which publishes short letters comprehensive articles reviews and perspectives in all aspects of biological and medicinal chemistry. Studies providing fundamental insights or describing novel syntheses as well as clinical or other applications-based work are welcomed.This broad scope includes experimental and theoretical studies on the chemical physical mechanistic and/or structural basis of biological or cell function in all domains of life. It encompasses the fields of chemical biology synthetic biology disease biology cell biology agriculture and food natural products research nucleic acid biology neuroscience structural biology and biophysics.The journal publishes studies that pertain to a broad range of medicinal chemistry including compound design and optimization biological evaluation molecular mechanistic understanding of drug delivery and drug delivery systems imaging agents and pharmacology and translational science of both small and large bioactive molecules. Novel computational cheminformatics and structural studies for the identification (or structure-activity relationship analysis) of bioactive molecules ligands and their targets are also welcome. The journal will consider computational studies applying established computational methods but only in combination with novel and original experimental data (e.g. in cases where new compounds have been designed and tested).Also included in the scope of the journal are articles relating to infectious diseases research on pathogens host-pathogen interactions therapeutics diagnostics vaccines drug-delivery systems and other biomedical technology development pertaining to infectious diseases.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


