Gene replacement therapy for spinal muscular atrophy: safety and preliminary efficacy in a Brazilian cohort
IF 4.6
3区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
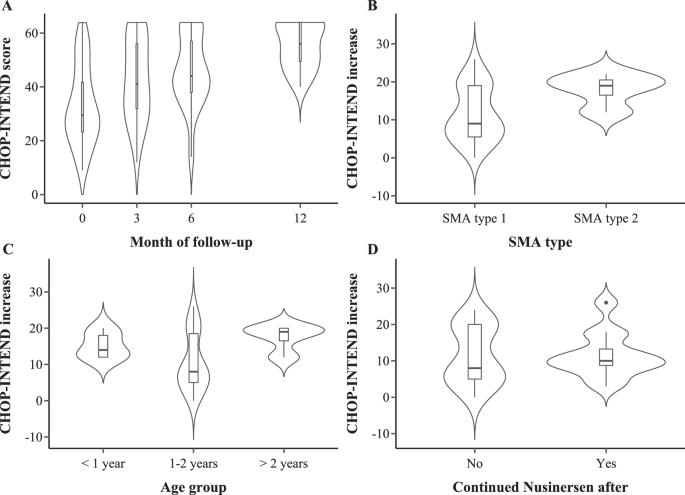
Spinal muscular atrophy (SMA) is a motor neuron disease associated with progressive muscle weakness, ventilatory failure, and reduced survival. Onasemnogene abeparvovec is the first gene replacement therapy (GT) approved to treat this condition. An observational retrospective study was conducted to assess adverse events and efficacy of GT in SMA patients. Forty-one patients with SMA (58.5% females and 80.1% SMA type 1) were included. The mean age at GT dosing was 18 (±6.4) months. Thirty-six patients (87.8%) were under previous treatment with nusinersen, and 10 (24.4%) continued nusinersen after GT. Mean CHOP-INTEND increased 13 points after 6 months and this finding did not differ between groups according to nusinersen maintenance after GT (p = 0.949). Among SMA type 1 patients, 14 (46.6%) reached the ability to sit alone. Liver transaminases elevation at least two times higher than the upper limit of normal value occurred in 29 (70.7%) patients. Thrombocytopenia occurred in 13 (31.7%) patients, and one presented thrombotic microangiopathy. Older age (>2 years) was associated with more prolonged use of corticosteroids (p = 0.021). GT is effective in SMA patients, combined nusinersen after GT did not appear to add gain in motor function and older age is associated with prolonged corticosteroid use.
脊髓性肌萎缩症的基因替代疗法:巴西队列的安全性和初步疗效。
脊髓性肌萎缩症(SMA)是一种运动神经元疾病,与进行性肌无力、呼吸衰竭和存活率降低有关。Onasemnogene abeparvovec是首个获准用于治疗这种疾病的基因替代疗法(GT)。我们开展了一项观察性回顾研究,以评估基因替代疗法对 SMA 患者的不良反应和疗效。研究纳入了 41 名 SMA 患者(58.5% 为女性,80.1% 为 SMA 1 型)。服用 GT 时的平均年龄为 18 (±6.4) 个月。36名患者(87.8%)曾接受过纽西奈森治疗,10名患者(24.4%)在GT治疗后继续接受纽西奈森治疗。6 个月后,CHOP-INTEND 的平均值增加了 13 分,根据 GT 后继续使用纽西奈森的情况,各组之间的结果并无差异(p = 0.949)。在 1 型 SMA 患者中,有 14 人(46.6%)能够单独坐立。29例(70.7%)患者的肝脏转氨酶高于正常值上限至少两倍。13例(31.7%)患者出现血小板减少,1例出现血栓性微血管病。年龄越大(大于 2 岁),使用皮质类固醇的时间越长(p = 0.021)。GT对SMA患者有效,但GT后联合使用纽西奈森似乎不会增加患者的运动功能,年龄越大,使用皮质类固醇的时间越长。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Gene Therapy
医学-生化与分子生物学
CiteScore
9.70
自引率
2.00%
发文量
67
审稿时长
4-8 weeks
期刊介绍:
Gene Therapy covers both the research and clinical applications of novel therapeutic techniques based on a genetic component. Over the last few decades, significant advances in technologies ranging from identifying novel genetic targets that cause disease through to clinical studies, which show therapeutic benefit, have elevated this multidisciplinary field to the forefront of modern medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


