HIF-1α and MIF enhance neutrophil-driven type 3 immunity and chondrogenesis in a murine spondyloarthritis model
IF 21.8
1区 医学
Q1 IMMUNOLOGY
引用次数: 0
Abstract
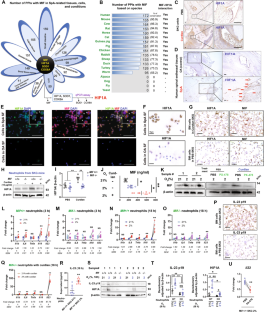
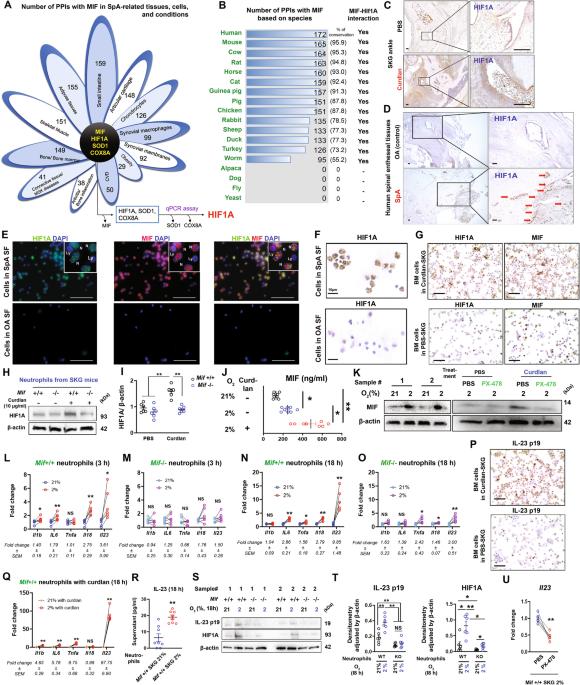
The hallmarks of spondyloarthritis (SpA) are type 3 immunity-driven inflammation and new bone formation (NBF). Macrophage migration inhibitory factor (MIF) was found to be a key driver of the pathogenesis of SpA by amplifying type 3 immunity, yet MIF-interacting molecules and networks remain elusive. Herein, we identified hypoxia-inducible factor-1 alpha (HIF1A) as an interacting partner molecule of MIF that drives SpA pathologies, including inflammation and NBF. HIF1A expression was increased in the joint tissues and synovial fluid of SpA patients and curdlan-injected SKG (curdlan-SKG) mice compared to the respective controls. Under hypoxic conditions in which HIF1A was stabilized, human and mouse neutrophils exhibited substantially increased expression of MIF and IL-23, an upstream type 3 immunity-related cytokine. Similar to MIF, systemic overexpression of IL-23 induced SpA pathology in SKG mice, while the injection of a HIF1A-selective inhibitor (PX-478) into curdlan-SKG mice prevented or attenuated SpA pathology, as indicated by a marked reduction in the expression of MIF and IL-23. Furthermore, genetic deletion of MIF or HIF1A inhibition with PX-478 in IL-23-overexpressing SKG mice did not induce evident arthritis or NBF, despite the presence of psoriasis-like dermatitis and blepharitis. We also found that MIF- and IL-23-expressing neutrophils infiltrated areas of the NBF in curdlan-SKG mice. These neutrophils potentially increased chondrogenesis and cell proliferation via the upregulation of STAT3 in periosteal cells and ligamental cells during endochondral ossification. Together, these results provide supporting evidence for an MIF/HIF1A regulatory network, and inhibition of HIF1A may be a novel therapeutic approach for SpA by suppressing type 3 immunity-mediated inflammation and NBF.
在小鼠脊柱关节炎模型中,HIF-1α 和 MIF 可增强中性粒细胞驱动的 3 型免疫和软骨生成作用
脊柱关节炎(Spondyloarthritis,SpA)的特征是3型免疫驱动的炎症和新骨形成(NBF)。研究发现,巨噬细胞迁移抑制因子(MIF)可通过放大3型免疫力而成为SpA发病机制的关键驱动因素,但与MIF相互作用的分子和网络仍然难以捉摸。在本文中,我们发现缺氧诱导因子-1α(HIF1A)是 MIF 的相互作用伙伴分子,它能驱动 SpA 的病理变化,包括炎症和 NBF。与对照组相比,SpA 患者和注射 curdlan 的 SKG(curdlan-SKG)小鼠的关节组织和滑液中 HIF1A 的表达均有所增加。在 HIF1A 被稳定的缺氧条件下,人和小鼠中性粒细胞的 MIF 和 IL-23(一种上游的 3 型免疫相关细胞因子)表达大幅增加。与 MIF 类似,IL-23 的系统性过表达也会诱发 SKG 小鼠的 SpA 病变,而向 curdlan-SKG 小鼠注射 HIF1A 选择性抑制剂(PX-478)则可防止或减轻 SpA 病变,这表现在 MIF 和 IL-23 的表达明显减少。此外,在 IL-23 高表达的 SKG 小鼠中遗传性缺失 MIF 或用 PX-478 抑制 HIF1A 不会诱发明显的关节炎或 NBF,尽管存在牛皮癣样皮炎和睑缘炎。我们还发现,表达 MIF- 和 IL-23 的中性粒细胞浸润了 curdlan-SKG 小鼠的 NBF 区域。在软骨内骨化过程中,这些中性粒细胞可能会通过上调骨膜细胞和韧带细胞中的 STAT3 增加软骨生成和细胞增殖。总之,这些结果为 MIF/HIF1A 调节网络提供了支持性证据,抑制 HIF1A 可抑制 3 型免疫介导的炎症和 NBF,从而成为治疗 SpA 的一种新方法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
31.20
自引率
1.20%
发文量
903
审稿时长
1 months
期刊介绍:
Cellular & Molecular Immunology, a monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, serves as a comprehensive platform covering both basic immunology research and clinical applications. The journal publishes a variety of article types, including Articles, Review Articles, Mini Reviews, and Short Communications, focusing on diverse aspects of cellular and molecular immunology.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


