Thermal decomposition kinetics and compatibility of NH3OHN5
引用次数: 0
Abstract
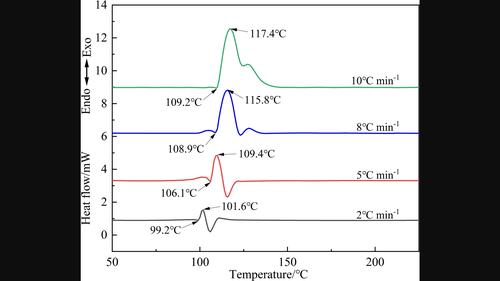
Hydroxylammonium cyclo‐pentazolate (NH3OHN5), as one of the poly‐nitrogen compounds, has a broad prospect in the field of energetic materials, due to its high specific impulse, high detonation velocity, and the pollution‐free products. In this paper, the thermal decomposition behavior of NH3OHN5 was studied by differential scanning calorimetry (DSC) using four heating rates (2, 5, 8, 10 °C min−1). The apparent activation energy (EK,O=114.31 kJ mol−1), the pre‐exponential factor (AK=4.78×1011 s−1) and the critical temperature of the thermal explosion (Tb=108.08 °C) of NH3OHN5 were calculated by Kissinger and Ozawa method under non‐isothermal heating conditions. The compatibility of NH3OHN5 with 1,3,5‐trinitro‐1,3,5‐triazacyclohexane (RDX), 1,3,5,7‐tetranitro‐1,3,5,7‐tetraazacyclooctane (HMX), 2,4,6,8,10,12‐hexanitro‐2,4,6,8,10,12‐hexaza‐isowurtzitane (CL‐20), ammonium perchlorate (AP), and hydroxy‐terminated polybutadiene (HTPB) were tested and judged based on a standard agreement (STANAG‐4147). The DSC results showed that NH3OHN5/HMX, NH3OHN5/RDX, NH3OHN5/CL‐20, NH3OHN5/AP and NH3OHN5/HTPB had good compatibility.
NH3OHN5 的热分解动力学和兼容性
羟基环五唑酸铵(NH3OHN5)作为多氮化合物之一,因其比冲大、爆速高、产物无污染等特点,在高能材料领域具有广阔的应用前景。本文采用差示扫描量热法(DSC)研究了 NH3OHN5 的热分解行为,使用了四种加热速率(2、5、8、10 °C min-1)。在非等温加热条件下,用基辛格和小泽法计算了 NH3OHN5 的表观活化能(EK,O=114.31 kJ mol-1)、预指数(AK=4.78×1011 s-1)和热爆炸临界温度(Tb=108.08 ℃)。NH3OHN5 与 1,3,5-三硝基-1,3,5-三氮杂环己烷(RDX)、1,3,5,7-四硝基-1,3,5,7-四氮杂环辛烷(HMX)、2,4,6,8,10,12-己硝基-2,4,6,8、10,12-hexanitro-2,4,6,8,10,12-hexaza-isowurtzitane (CL-20)、高氯酸铵 (AP) 和羟基封端聚丁二烯 (HTPB) 进行了测试,并根据标准协议(STANAG-4147)进行了评判。DSC 结果表明,NH3OHN5/HMX、NH3OHN5/RDX、NH3OHN5/CL-20、NH3OHN5/AP 和 NH3OHN5/HTPB 具有良好的相容性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


