Multicenter assessment and longitudinal study of the prevalence of antibodies and related adaptive immune responses to AAV in adult males with hemophilia
IF 4.6
3区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
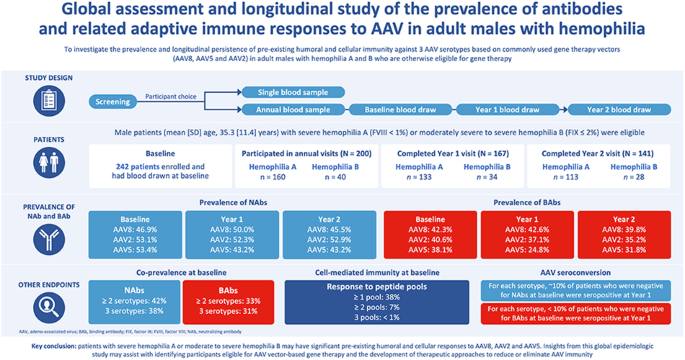
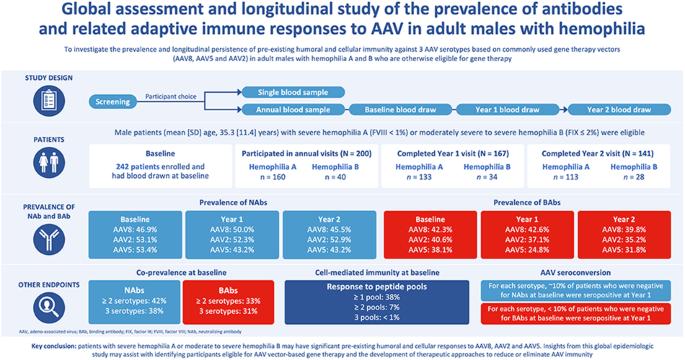
Adeno-associated virus (AAV) based gene therapy has demonstrated effective disease control in hemophilia. However, pre-existing immunity from wild-type AAV exposure impacts gene therapy eligibility. The aim of this multicenter epidemiologic study was to determine the prevalence and persistence of preexisting immunity against AAV2, AAV5, and AAV8, in adult participants with hemophilia A or B. Blood samples were collected at baseline and annually for ≤3 years at trial sites in Austria, France, Germany, Italy, Spain, and the United States. At baseline, AAV8, AAV2, and AAV5 neutralizing antibodies (NAbs) were present in 46.9%, 53.1%, and 53.4% of participants, respectively; these values remained stable at Years 1 and 2. Co-prevalence of NAbs to at least two serotypes and all three serotypes was present at baseline for ~40% and 38.2% of participants, respectively. For each serotype, ~10% of participants who tested negative for NAbs at baseline were seropositive at Year 1. At baseline, 38.3% of participants had detectable cell mediated immunity by ELISpot, although no correlations were observed with the humoral response. In conclusion, participants with hemophilia may have significant preexisting immunity to AAV capsids. Insights from this study may assist in understanding capsid-based immunity trends in participants considering AAV vector-based gene therapy.
多中心评估和纵向研究成年男性血友病患者对 AAV 的抗体和相关适应性免疫反应的流行情况。
基于腺相关病毒(AAV)的基因疗法已证明能有效控制血友病的病情。然而,野生型 AAV 暴露产生的原有免疫力会影响基因治疗的资格。这项多中心流行病学研究旨在确定 AAV2、AAV5 和 AAV8 在成年 A 型或 B 型血友病患者中的普遍性和持续性。基线时,分别有46.9%、53.1%和53.4%的参与者体内存在AAV8、AAV2和AAV5中和抗体(NAbs);这些数值在第一年和第二年保持稳定。至少两种血清型和所有三种血清型的 NAbs 共患病率在基线时分别为 40% 和 38.2%。对于每种血清型,基线时NAbs检测呈阴性的参与者中约有10%在第一年时血清呈阳性。在基线时,38.3% 的参与者通过 ELISpot 检测到细胞介导免疫,但没有观察到与体液反应的相关性。总之,血友病患者可能对 AAV 病毒衣壳有明显的预先免疫力。这项研究的启示可能有助于了解考虑使用 AAV 载体进行基因治疗的参与者的囊壳免疫趋势。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Gene Therapy
医学-生化与分子生物学
CiteScore
9.70
自引率
2.00%
发文量
67
审稿时长
4-8 weeks
期刊介绍:
Gene Therapy covers both the research and clinical applications of novel therapeutic techniques based on a genetic component. Over the last few decades, significant advances in technologies ranging from identifying novel genetic targets that cause disease through to clinical studies, which show therapeutic benefit, have elevated this multidisciplinary field to the forefront of modern medicine.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


