Pannexin-3 stabilizes the transcription factor Bcl6 in a channel-independent manner to protect against vascular oxidative stress
IF 6.7
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Targeted degradation regulates the activity of the transcriptional repressor Bcl6 and its ability to suppress oxidative stress and inflammation. Here, we report that abundance of endothelial Bcl6 is determined by its interaction with Golgi-localized pannexin 3 (Panx3) and that Bcl6 transcriptional activity protects against vascular oxidative stress. Consistent with data from obese, hypertensive humans, mice with an endothelial cell–specific deficiency in Panx3 had spontaneous systemic hypertension without obvious changes in channel function, as assessed by Ca2+ handling, ATP amounts, or Golgi luminal pH. Panx3 bound to Bcl6, and its absence reduced Bcl6 protein abundance, suggesting that the interaction with Panx3 stabilized Bcl6 by preventing its degradation. Panx3 deficiency was associated with increased expression of the gene encoding the H2O2-producing enzyme Nox4, which is normally repressed by Bcl6, resulting in H2O2-induced oxidative damage in the vasculature. Catalase rescued impaired vasodilation in mice lacking endothelial Panx3. Administration of a newly developed peptide to inhibit the Panx3-Bcl6 interaction recapitulated the increase in Nox4 expression and in blood pressure seen in mice with endothelial Panx3 deficiency. Panx3-Bcl6-Nox4 dysregulation occurred in obesity-related hypertension, but not when hypertension was induced in the absence of obesity. Our findings provide insight into a channel-independent role of Panx3 wherein its interaction with Bcl6 determines vascular oxidative state, particularly under the adverse conditions of obesity.
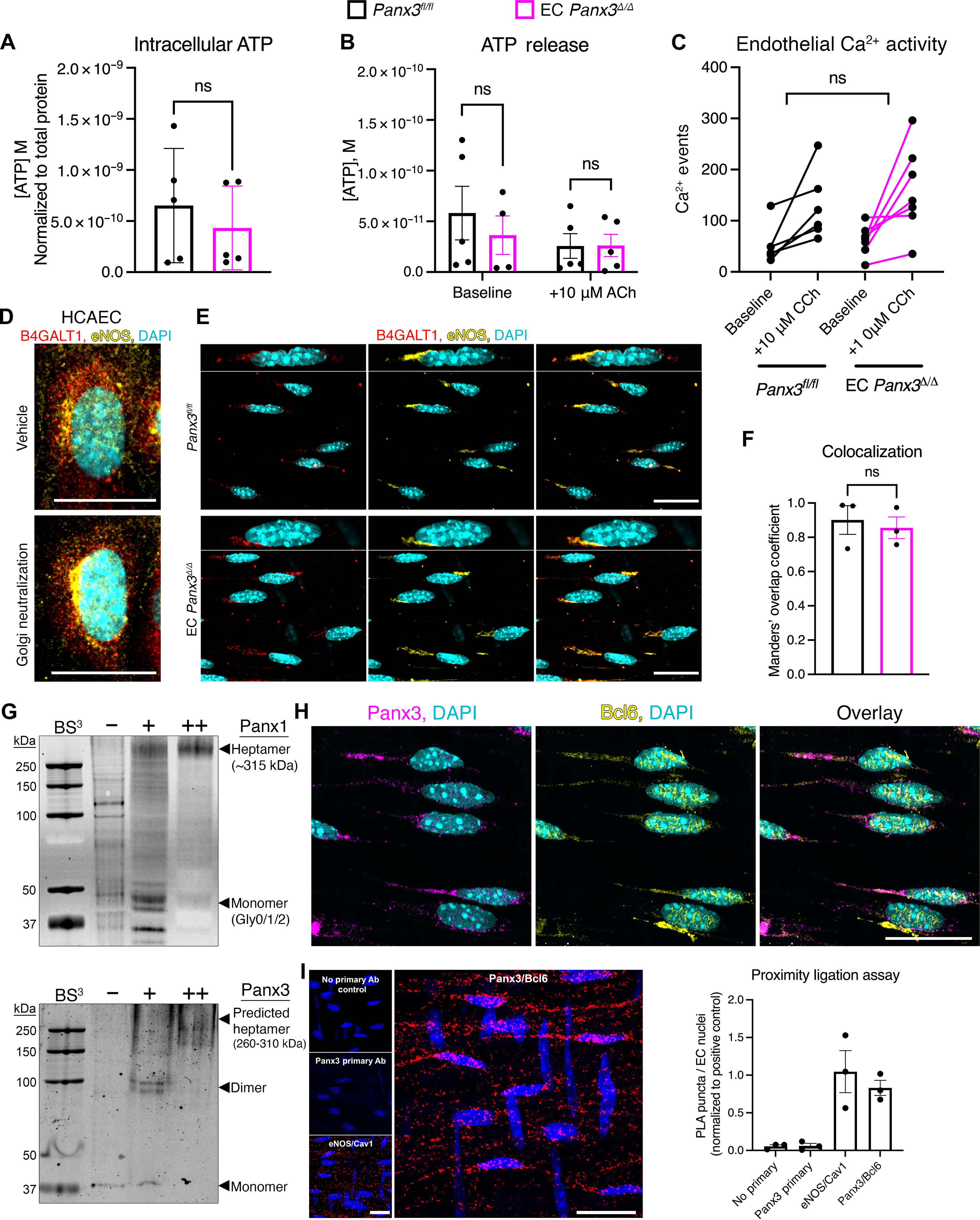
Pannexin-3 以一种与通道无关的方式稳定转录因子 Bcl6,从而保护血管免受氧化应激。
靶向降解调节转录抑制因子 Bcl6 的活性及其抑制氧化应激和炎症的能力。在这里,我们报告了内皮细胞 Bcl6 的丰度是由其与定位在高尔基体的 Pannexin 3(Panx3)的相互作用决定的,而且 Bcl6 的转录活性能保护血管免受氧化应激。与肥胖、高血压患者的数据一致,内皮细胞特异性缺乏 Panx3 的小鼠会出现自发性系统性高血压,但通道功能没有明显变化,这是由 Ca2+ 处理、ATP 量或高尔基体腔 pH 值评估得出的结果。Panx3与Bcl6结合,其缺失会降低Bcl6蛋白的丰度,这表明与Panx3的相互作用通过阻止Bcl6降解而使其稳定。Panx3的缺乏与编码H2O2生成酶Nox4的基因表达增加有关,而Nox4通常被Bcl6抑制,从而导致H2O2诱导的血管氧化损伤。过氧化氢酶能挽救缺乏内皮细胞 Panx3 的小鼠受损的血管舒张功能。服用一种新开发的抑制 Panx3-Bcl6 相互作用的多肽,可再现内皮 Panx3 缺乏小鼠体内 Nox4 表达和血压的增加。Panx3-Bcl6-Nox4失调发生在与肥胖相关的高血压中,而在没有肥胖的情况下诱发高血压时则不会发生。我们的研究结果使我们深入了解了 Panx3 在与 Bcl6 的相互作用中决定血管氧化状态的一种独立于通道的作用,尤其是在肥胖的不利条件下。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Science Signaling
BIOCHEMISTRY & MOLECULAR BIOLOGY-CELL BIOLOGY
CiteScore
9.50
自引率
0.00%
发文量
148
审稿时长
3-8 weeks
期刊介绍:
"Science Signaling" is a reputable, peer-reviewed journal dedicated to the exploration of cell communication mechanisms, offering a comprehensive view of the intricate processes that govern cellular regulation. This journal, published weekly online by the American Association for the Advancement of Science (AAAS), is a go-to resource for the latest research in cell signaling and its various facets.
The journal's scope encompasses a broad range of topics, including the study of signaling networks, synthetic biology, systems biology, and the application of these findings in drug discovery. It also delves into the computational and modeling aspects of regulatory pathways, providing insights into how cells communicate and respond to their environment.
In addition to publishing full-length articles that report on groundbreaking research, "Science Signaling" also features reviews that synthesize current knowledge in the field, focus articles that highlight specific areas of interest, and editor-written highlights that draw attention to particularly significant studies. This mix of content ensures that the journal serves as a valuable resource for both researchers and professionals looking to stay abreast of the latest advancements in cell communication science.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


