A one-pot thiomethylation of pyrrole and indoles
IF 1
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
C(sp2)H functionalization of pyrrole, indole, indolyl-3-acetic acid (heteroauxin), and 2-amino-3-(indol-3yl)propionic acid (tryptophan) was carried out via a one-pot C-thiomethylation with formaldehyde and mercaptans in pyridine which simultaneously played the role of a catalyst and solvent. The possibility of the synthesis of structurally diverse sulfanyl methylindoles depending on the nature of the reagents was demonstrated.
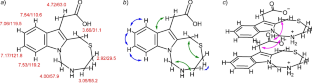
吡咯和吲哚的一锅硫代甲基化反应
在同时起催化剂和溶剂作用的吡啶中,通过与甲醛和硫醇的单锅 C-硫甲基化反应,对吡咯、吲哚、吲哚基-3-乙酸(杂华素)和 2-氨基-3-(吲哚基-3)丙酸(色氨酸)进行了 C(sp2)H 功能化。实验证明,根据试剂的性质,可以合成结构多样的硫代甲基吲哚。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
2.90
自引率
13.30%
发文量
98
审稿时长
1 months
期刊介绍:
The international journal Chemistry of Heterocyclic Compounds publishes original papers, short communications, reviews, and mini-reviews dealing with problems in the field of heterocyclic chemistry in Russian and English. The Journal also publishes reviews and annotations on new books and brief reports on conferences in the field of heterocyclic chemistry, as well as commemoratives dedicated to prominent heterocyclic chemists.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


