Influence of contrasting redox conditions on iron (oxyhydr)oxide transformation and associated phosphate sorption
Abstract
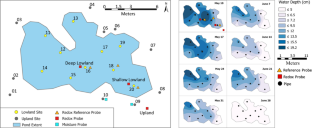
Iron (oxyhydr)oxides strongly adsorb phosphate and limit its bioavailability, but interactions between phosphate and various Fe (oxyhydr)oxides are poorly constrained in natural systems. An in-situ incubation experiment was conducted to explore Fe (oxyhydr)oxide transformation and effects on phosphate sorption in soils with contrasting saturation and redox conditions. Synthetic Fe (oxyhydr)oxides (ferrihydrite, goethite and hematite) were coated onto quartz sand and either pre-sorbed with phosphate or left phosphate-free. The oxide-coated sands were mixed with natural organic matter, enclosed in mesh bags, and buried in and around a vernal pond for up to 12 weeks. Redox conditions were stable and oxic in the upland soils surrounding the vernal pond but largely shifted from Fe reducing to Fe oxidizing in the lowland soils within the vernal pond as it dried during the summer. Iron (oxyhydr)oxides lost more Fe (− 41% ± 10%) and P (− 43 ± 11%) when incubated in the redox-dynamic lowlands compared to the uplands (− 18% ± 5% Fe and − 24 ± 8% P). Averaged across both uplands and lowlands, Fe losses from crystalline goethite and hematite (− 38% ± 6%) were unexpectedly higher than losses from short range ordered ferrihydrite (− 12% ± 10%). We attribute losses of Fe and associated P from goethite and hematite to colloid detachment and dispersion but losses from ferrihydrite to reductive dissolution. Iron losses were partially offset by retention of solubilized Fe as organic-bound Fe(III). Iron (oxyhydr)oxides that persisted during the incubation retained or even gained P, indicating low amounts of phosphate sorption from solution. These results demonstrate that hydrologic variability and Fe (oxyhydr)oxide mineralogy impact Fe mobilization pathways that may regulate phosphate bioavailability.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


