A non-diazo approach to functionalized (2-furyl)-2-pyrrolidines through a cascade reaction of enynal-derived zinc carbenoids with β-arylaminoketones†
IF 4.6
1区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
This study introduces a cascade approach for synthesizing functionalized (2-furyl)-2-pyrrolidines, showcasing both convergence and remarkable stereoselectivity. This domino process proceeds through an N–H insertion into an enynal-derived metal–carbenoid, followed by an intramolecular aldol reaction to provide pyrrolidines with high diastereoselectivity (98 : 2). This chemistry utilizes Earth-abundant zinc chloride as a catalyst with loading as low as 1 mol%. This method operates under mild conditions and demonstrates high chemoselectivity by accommodating substrates bearing functionalities such as free alcohols, alkenes, and alkynes.
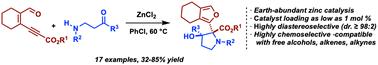
烯醛衍生的类锌与β-芳胺酮级联反应制备功能化(2-呋喃基)-2-吡咯烷的非重氮方法
本研究介绍了一种级联合成功能化(2-呋喃基)-2-吡咯烷的方法,该方法具有收敛性和显著的立体选择性。这个多米诺骨牌过程是通过N-H插入到乙烯衍生的金属-类碳化合物中,然后是…
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic Chemistry Frontiers
CHEMISTRY, ORGANIC-
CiteScore
7.90
自引率
11.10%
发文量
686
审稿时长
1 months
期刊介绍:
Organic Chemistry Frontiers is an esteemed journal that publishes high-quality research across the field of organic chemistry. It places a significant emphasis on studies that contribute substantially to the field by introducing new or significantly improved protocols and methodologies. The journal covers a wide array of topics which include, but are not limited to, organic synthesis, the development of synthetic methodologies, catalysis, natural products, functional organic materials, supramolecular and macromolecular chemistry, as well as physical and computational organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


