Diastereoselective Synthesis of Carbohydrate Conjugates: Pyrano[3,2-c]quinolones
IF 2.2
4区 化学
Q2 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Abstract A facile and efficient protocol for the diastereoselective synthesis of pyrano[3,2-c]quinolone carbohydrate derivatives from Perlin aldehydes and 4-hydroxyquinolones has been developed using a one-pot condensation at room temperature. In this investigation, glucose and galactose were employed as inexpensive starting materials to synthesize two sets of pyrano[3,2-c]quinolone-based carbohydrate conjugates. A total of sixteen novel compounds were successfully synthesized using this methodology in good to excellent yields. The reaction exhibited remarkable diastereoselectivity, resulting in a single diastereomeric product with a diastereomeric excess (dr) 97:3 for glucose, while a diastereomeric mixture with a diastereomeric excess (dr) 67:33 was obtained for galactose. The structural characterization of all sixteen compounds was carried out using various analytical techniques, including IR, 1H NMR, 13C NMR, 1H-1H COSY, 1H-13C HETCOR experiments, 2D NOESY NMR, and HRMS data. Additionally, the scalability of the protocol was successfully demonstrated by synthesizing one of the compounds on a gram scale, highlighting its potential for large-scale production.
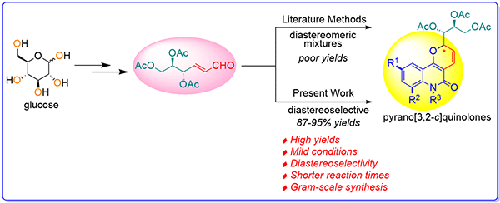
碳水化合物偶联物的非对映选择性合成:吡喃[3,2-c]喹诺酮类
摘要:本文研究了一种在室温条件下由柏林醛和4-羟基喹诺酮类化合物合成吡喃[3,2-c]喹诺酮类化合物的非对映选择性反应。本研究以葡萄糖和半乳糖为原料合成了两组吡喃[3,2-c]喹诺酮类碳水化合物缀合物。利用该方法成功合成了16个新化合物,收率较高。该反应表现出显著的非对映选择性,葡萄糖的非对映异构体过量(dr)为97:3,而半乳糖的非对映异构体过量(dr)为67:33。所有16个化合物的结构表征使用各种分析技术进行,包括IR, 1H NMR, 13C NMR, 1H COSY, 1H-13C HETCOR实验,2D NOESY NMR和HRMS数据。此外,通过在克尺度上合成一种化合物,成功地证明了该协议的可扩展性,突出了其大规模生产的潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Synthesis-Stuttgart
化学-有机化学
CiteScore
4.50
自引率
7.70%
发文量
435
审稿时长
1 months
期刊介绍:
SYNTHESIS is an international full-paper journal devoted to the advancement of the science of chemical synthesis. It covers all fields of organic chemistry involving synthesis, including catalysis, organometallic, medicinal, biological, and photochemistry, but also related disciplines. SYNTHESIS provides dependable research results with detailed and reliable experimental procedures and full characterization of all important new products as well as scientific primary data.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


