Convenient Synthesis of Functionalized Tetracyclic Dihydrochromeno[2,3-b]pyrrolo[3,4-e]pyridine-triones via Four-Component Reactions
IF 2.3
4区 化学
Q2 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Abstract In this research, a simple and efficient strategy for the straightforward synthesis of tetracyclic dihydrochromeno[2,3-b]pyrrolo[3,4-e]pyridine-trione derivatives is presented by a sequential four-component reaction of arylamines, dimethyl acetylenedicarboxylate, alkylamines, and 3-formylchromones as readily available starting materials in MeOH at room temperature. The merit of this sequential enamine formation/1,2-nucleophilic addition/intramolecular aza-Michael addition is highlighted by its high energy conserving (short reaction times at room temperature), excellent yields, metal-free catalyst, easy purification (the products can be purified by simple filtration and washing with EtOH), and green and mild conditions in a one-pot reaction. Presumably, there are no previous reports for the synthesis of these classes of heterocyclic dihydrochromeno[2,3-b]pyrrolo[3,4-e]pyridine-trione derivatives.
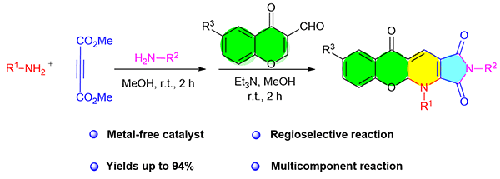
四组分反应合成功能化四环二氢色[2,3-b]吡咯[3,4-e]吡啶三酮
摘要本研究提出了一种简单高效的方法,以芳胺、二甲基乙基二羧酸酯、烷基胺和3-甲酰色酮为原料,在室温条件下在甲醇中进行四组分连续反应,直接合成四环二氢铬[2,3-b]吡咯[3,4-e]吡啶-三酮衍生物。这种顺序形成烯胺/1,2-亲核加成/分子内aza-Michael加成的优点是高节能(室温下反应时间短)、产率高、无金属催化剂、易于纯化(产物可以通过简单的过滤和EtOH洗涤纯化)、绿色和温和的一锅反应条件。据推测,这类杂环二氢色[2,3-b]吡咯[3,4-e]吡啶-三酮衍生物的合成尚未见报道。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Synthesis-Stuttgart
化学-有机化学
CiteScore
4.50
自引率
7.70%
发文量
435
审稿时长
1 months
期刊介绍:
SYNTHESIS is an international full-paper journal devoted to the advancement of the science of chemical synthesis. It covers all fields of organic chemistry involving synthesis, including catalysis, organometallic, medicinal, biological, and photochemistry, but also related disciplines. SYNTHESIS provides dependable research results with detailed and reliable experimental procedures and full characterization of all important new products as well as scientific primary data.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



