Genes ingenuity pathway analysis unveils smoothelin‐like 1 (SMTNL1 ) as a key regulatory protein involved in sodium pentobarbital‐induced growth inhibition in breast cancer
IF 2.9
4区 医学
Q2 PHARMACOLOGY & PHARMACY
引用次数: 0
Abstract
Abstract We previously reported that sodium pentobarbital inhibited the growth of the breast cancer associated with the normalization of microcirculatory hemodynamics and oxygenation. Here, we aimed to screen the key regulatory proteins involved in pentobarbital‐induced normalization of microcirculatory hemodynamics in the breast cancer tissues. A nude mice model of xenograft was established using triple negative breast cancer cell line MDA‐MB‐231. After tumor cell implantation, the mice were subcutaneously injected with 50 mg/kg/day of sodium pentobarbital or an equal volume of solvent adjacent to the tumor for 14 days. Liquid chromatography linked to tandem mass spectrometry (LC–MS/MS) was used to analyze the difference in protein expression profile between the two groups. Ingenuity pathway analysis (IPA) was used to perform the canonical pathway analysis, upstream regulators analysis, and protein–protein interaction networks analysis. Screened proteins were confirmed by real‐time quantitative polymerase chain reaction (RT–qPCR) and Western blot analysis. A total of 101 differentially expressed proteins were revealed between groups. Canonical pathway analysis suggested that acute phase response signaling ( z = 1, p = .00208), dilated cardiomyopathy signaling pathway ( z = −2, p = .00671), and ILK signaling ( z = 1, p = .0172) were key pathways with highlight associations. The mRNA and protein expressions of SMTNL1 were found significantly decreased in pentobarbital‐treated tumor tissues compared with those in controls (both p < .01). Nine important protein–protein interaction networks were identified, and of which, two contained multiple downstream regulatory proteins of SMTNL1. In conclusion, SMTNL1 is revealed as a key protein involved in pentobarbital‐induced growth inhibition signaling in breast cancer. SMTNL1 may become a new potential target for tumor microcirculation research.
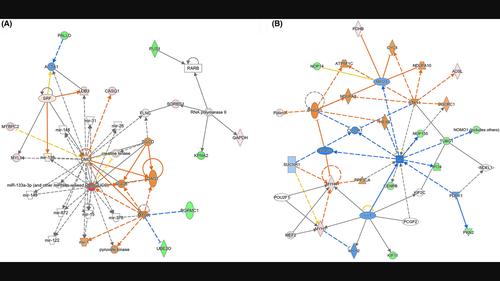
基因独创性通路分析揭示SMTNL1是一个关键的调节蛋白,参与戊巴比妥钠诱导的乳腺癌生长抑制
我们之前报道过戊巴比妥钠抑制与微循环血流动力学和氧合正常化相关的乳腺癌生长。在这里,我们旨在筛选戊巴比妥诱导乳腺癌组织微循环血流动力学正常化的关键调节蛋白。采用三阴性乳腺癌细胞系MDA‐MB‐231建立了裸鼠异种移植瘤模型。肿瘤细胞植入后,小鼠在肿瘤旁皮下注射戊巴比妥钠50 mg/kg/d或等体积的溶剂,持续14 d。采用液相色谱-串联质谱(LC-MS /MS)分析两组蛋白表达谱的差异。匠心途径分析(Ingenuity pathway analysis, IPA)用于典型途径分析、上游调控因子分析和蛋白质相互作用网络分析。筛选的蛋白通过实时定量聚合酶链反应(RT-qPCR)和Western blot分析证实。组间共发现101个差异表达蛋白。典型信号通路分析表明,急性期反应信号通路(z = 1, p = 0.00208)、扩张型心肌病信号通路(z = - 2, p = 0.00671)和ILK信号通路(z = 1, p = 0.0172)是具有突出相关性的关键信号通路。与对照组相比,戊巴比妥治疗的肿瘤组织中SMTNL1的mRNA和蛋白表达均显著降低(p <. 01)。鉴定出9个重要的蛋白-蛋白相互作用网络,其中2个含有SMTNL1的多个下游调节蛋白。综上所述,SMTNL1是参与戊巴比妥诱导的乳腺癌生长抑制信号传导的关键蛋白。SMTNL1可能成为肿瘤微循环研究新的潜在靶点。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Pharmacology Research & Perspectives
Pharmacology, Toxicology and Pharmaceutics-General Pharmacology, Toxicology and Pharmaceutics
CiteScore
5.30
自引率
3.80%
发文量
120
审稿时长
20 weeks
期刊介绍:
PR&P is jointly published by the American Society for Pharmacology and Experimental Therapeutics (ASPET), the British Pharmacological Society (BPS), and Wiley. PR&P is a bi-monthly open access journal that publishes a range of article types, including: target validation (preclinical papers that show a hypothesis is incorrect or papers on drugs that have failed in early clinical development); drug discovery reviews (strategy, hypotheses, and data resulting in a successful therapeutic drug); frontiers in translational medicine (drug and target validation for an unmet therapeutic need); pharmacological hypotheses (reviews that are oriented to inform a novel hypothesis); and replication studies (work that refutes key findings [failed replication] and work that validates key findings). PR&P publishes papers submitted directly to the journal and those referred from the journals of ASPET and the BPS
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


