One year of experience with combined pharmacokinetic/pharmacogenetic monitoring of anti-TNF alpha agents: a retrospective study
IF 2.9
3区 医学
Q2 GENETICS & HEREDITY
引用次数: 0
Abstract
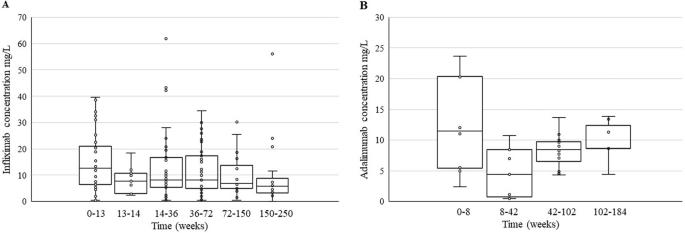
Anti-tumor necrosis factor alpha (anti-TNFα) inhibitors are used extensively for the management of moderate to severe inflammatory bowel disease (IBD) in both adult and pediatric patients. Unfortunately, not all patients show an optimal response to induction therapy, while others lose their response over time for reasons yet poorly understood. We report on a pharmacokinetic/pharmacogenetic approach to monitor the therapy with anti-TNFα in a real-world cohort of seventy-nine pediatric patients affected by IBD that was analyzed retrospectively. We evaluated plasma concentrations of infliximab, adalimumab, and related anti-drug antibodies (ADAs), and single nucleotide polymorphisms (SNPs) in genes involved in immune processes and inflammation on the anti-TNFα response. We found a significant association between the SNP in TNFα promoter (−308G>A) and clinical remission without steroids in patients on infliximab therapy. Additionally, a potential connection between HLA-DQA1*05 genetic variant carriers and a higher risk of anti-TNFα immunogenicity emerged.
抗TNF-α药物药代动力学/药理学联合监测一年的经验:一项回顾性研究。
抗肿瘤坏死因子-α(Anti-TNFα)抑制剂广泛用于成人和儿童中重度炎症性肠病(IBD)的治疗。不幸的是,并非所有患者都对诱导治疗表现出最佳反应,而其他患者则因尚不清楚的原因而随着时间的推移而失去反应。我们报告了一种药代动力学/药物遗传学方法,在一个由79名IBD患儿组成的真实队列中监测抗TNFα的治疗,该队列进行了回顾性分析。我们评估了英夫利昔单抗、阿达木单抗和相关抗药物抗体(ADAs)的血浆浓度,以及参与免疫过程和炎症的基因对抗TNFα反应的单核苷酸多态性(SNPs)。我们发现,在接受英夫利昔单抗治疗的患者中,TNFα启动子中的SNP(-308G>a)与无类固醇的临床缓解之间存在显著关联。此外,HLA-DQA1*05基因变异携带者与抗TNFα免疫原性的更高风险之间存在潜在联系。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Pharmacogenomics Journal
医学-药学
CiteScore
7.20
自引率
0.00%
发文量
35
审稿时长
6-12 weeks
期刊介绍:
The Pharmacogenomics Journal is a print and electronic journal, which is dedicated to the rapid publication of original research on pharmacogenomics and its clinical applications.
Key areas of coverage include:
Personalized medicine
Effects of genetic variability on drug toxicity and efficacy
Identification and functional characterization of polymorphisms relevant to drug action
Pharmacodynamic and pharmacokinetic variations and drug efficacy
Integration of new developments in the genome project and proteomics into clinical medicine, pharmacology, and therapeutics
Clinical applications of genomic science
Identification of novel genomic targets for drug development
Potential benefits of pharmacogenomics.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


