Demystifying the impact of prenatal tobacco exposure on the placental immune microenvironment: Avoiding the tragedy of mending the fold after death
IF 4.2
2区 医学
Q1 Biochemistry, Genetics and Molecular Biology
引用次数: 0
Abstract
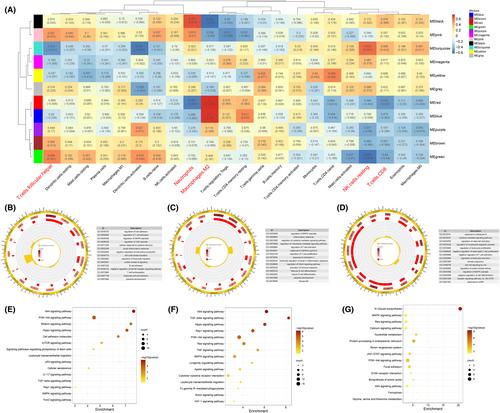
Prenatal tobacco exposure (PTE) correlates significantly with a surge in adverse pregnancy outcomes, yet its pathological mechanisms remain partially unexplored. This study aims to meticulously examine the repercussions of PTE on placental immune landscapes, employing a coordinated research methodology encompassing bioinformatics, machine learning and animal studies. Concurrently, it aims to screen biomarkers and potential compounds that could sensitively indicate and mitigate placental immune disorders. In the course of this research, two gene expression omnibus (GEO) microarrays, namely GSE27272 and GSE7434, were included. Gene set enrichment analysis (GSEA) and immune enrichment investigations on differentially expressed genes (DEGs) indicated that PTE might perturb numerous innate or adaptive immune‐related biological processes. A cohort of 52 immune‐associated DEGs was acquired by cross‐referencing the DEGs with gene sets derived from the ImmPort database. A protein–protein interaction (PPI) network was subsequently established, from which 10 hub genes were extracted using the maximal clique centrality (MCC) algorithm (JUN, NPY, SST, FLT4, FGF13, HBEGF, NR0B2, AREG, NR1I2, SEMA5B). Moreover, we substantiated the elevated affinity of tobacco reproductive toxicants, specifically nicotine and nitrosamine, with hub genes through molecular docking (JUN, FGF13 and NR1I2). This suggested that these genes could potentially serve as crucial loci for tobacco's influence on the placental immune microenvironment. To further elucidate the immune microenvironment landscape, consistent clustering analysis was conducted, yielding three subtypes, where the abundance of follicular helper T cells (p < 0.05) in subtype A, M2 macrophages (p < 0.01), neutrophils (p < 0.05) in subtype B and CD8+ T cells (p < 0.05), resting NK cells (p < 0.05), M2 macrophages (p < 0.05) in subtype C were significantly different from the control group. Additionally, three pivotal modules, designated as red, blue and green, were identified, each bearing a close association with differentially infiltrated immunocytes, as discerned by the weighted gene co‐expression network analysis (WGCNA). Functional enrichment analysis was subsequently conducted on these modules. To further probe into the mechanisms by which immune‐associated DEGs are implicated in intercellular communication, 20 genes serving as ligands or receptors and connected to differentially infiltrating immunocytes were isolated. Employing a variety of machine learning techniques, including one‐way logistic regression, LASSO regression, random forest and artificial neural networks, we screened 11 signature genes from the intersection of immune‐associated DEGs and secretory protein‐encoding genes derived from the Human Protein Atlas. Notably, CCL18 and IFNA4 emerged as prospective peripheral blood markers capable of identifying PTE‐induced immune disorders. These markers demonstrated impressive predictive power, as indicated by the area under the curve (AUC) of 0.713 (0.548–0.857) and 0.780 (0.618–0.914), respectively. Furthermore, we predicted 34 potential compounds, including cyclosporine, oestrogen and so on, which may engage with hub genes and attenuate immune disorders instigated by PTE. The diagnostic performance of these biomarkers, alongside the interventional effect of cyclosporine, was further corroborated in animal studies via ELISA, Western blot and immunofluorescence assays. In summary, this study identifies a disturbance in the placental immune landscape, a secondary effect of PTE, which may underlie multiple pregnancy complications. Importantly, our research contributes to the noninvasive and timely detection of PTE‐induced placental immune disorders, while also offering innovative therapeutic strategies for their treatment.
解开产前烟草暴露对胎盘免疫微环境的影响:避免死后补褶的悲剧。
产前烟草暴露(PTE)与不良妊娠结局的激增显著相关,但其病理机制仍部分未被探索。本研究旨在采用包括生物信息学、机器学习和动物研究在内的协调研究方法,仔细研究PTE对胎盘免疫景观的影响。同时,它旨在筛选能够敏感地指示和减轻胎盘免疫障碍的生物标志物和潜在化合物。在本研究过程中,纳入了两个基因表达综合(GEO)微阵列,即GSE27272和GSE7434。基因集富集分析(GSEA)和对差异表达基因(DEG)的免疫富集研究表明,PTE可能干扰许多先天或适应性免疫相关的生物学过程。通过将DEG与来自ImmPort数据库的基因集交叉引用,获得了52个免疫相关DEG的队列。随后建立了蛋白质-蛋白质相互作用(PPI)网络,使用最大团中心性(MCC)算法(JUN、NPY、SST、FLT4、FGF13、HBEGF、NR0B2、AREG、NR1I2、SEMA5B)从中提取10个枢纽基因。此外,我们通过分子对接证实了烟草生殖毒物,特别是尼古丁和亚硝胺,与中枢基因(JUN、FGF13和NR1I2)的亲和力升高。这表明,这些基因可能是烟草影响胎盘免疫微环境的关键基因座。为了进一步阐明免疫微环境景观,进行了一致的聚类分析,产生了三种亚型,其中卵泡辅助T细胞的丰度(p
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
10.00
自引率
1.90%
发文量
496
审稿时长
28 weeks
期刊介绍:
Bridging physiology and cellular medicine, and molecular biology and molecular therapeutics, Journal of Cellular and Molecular Medicine publishes basic research that furthers our understanding of the cellular and molecular mechanisms of disease and translational studies that convert this knowledge into therapeutic approaches.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


