Norah A Owiti, Simran Kaushal, Lincoln Martin, Jamie Sly, Carol D Swartz, Jasmine Fowler, Joshua J Corrigan, Les Recio, Bevin P Engelward
{"title":"Using the HepaCometChip Assay for Broad-Spectrum DNA Damage Analysis.","authors":"Norah A Owiti, Simran Kaushal, Lincoln Martin, Jamie Sly, Carol D Swartz, Jasmine Fowler, Joshua J Corrigan, Les Recio, Bevin P Engelward","doi":"10.1002/cpz1.563","DOIUrl":null,"url":null,"abstract":"<p><p>Exposure to DNA damaging agents can lead to mutations that cause cancer. The liver is particularly vulnerable because it contains high levels of Cytochrome P450 enzymes that can convert xenobiotics into DNA reactive metabolites that form potentially carcinogenic bulky DNA adducts. As such, current requirements for preclinical testing include in vivo testing for DNA damage in the liver, which often requires many animals. Given that efforts are underway in many countries to reduce or eliminate the use of animals in research, there is a critical need for fast and robust in vitro tests to discern whether xenobiotics or potential pharmaceutical agents can damage the hepatocyte genome. One possible approach is to leverage the alkaline comet assay, which is used to assess genotoxicity based on the ability of damaged DNA to become free to migrate toward the anode during electrophoresis. The comet assay, however, has several limitations. The assay is (i) slow and (ii) vulnerable to experimental noise, (iii) it is difficult to detect bulky DNA adducts since they do not directly affect DNA migration, and (iv) cell types typically used do not have robust metabolic capacity. To address some of these concerns, we have developed the \"HepaCometChip\" (a.k.a. the HepaRG CometChip), wherein metabolically competent cells are incorporated into a higher throughput CometChip platform. Repair trapping is used to increase sensitivity for bulky lesions: undetectable bulky lesions are converted into repair intermediates (specifically, single-strand breaks) that can be detected with the assay. Here, we describe a protocol for performing the HepaCometChip assay that includes handling and dosing of HepaRG cells and performing the CometChip assay. With its higher throughput, ability to capture metabolic activation, and sensitivity to bulky lesions, the HepaCometChip offers a potential alternative to the use of animals for genotoxicity testing. © 2022 The Authors. Current Protocols published by Wiley Periodicals LLC. Basic Protocol 1: HepaRG cell culturing and dosing Basic Protocol 2: CometChip assay.</p>","PeriodicalId":11174,"journal":{"name":"Current Protocols","volume":"2 9","pages":"e563"},"PeriodicalIF":0.0000,"publicationDate":"2022-09-01","publicationTypes":"Journal Article","fieldsOfStudy":null,"isOpenAccess":false,"openAccessPdf":"https://ftp.ncbi.nlm.nih.gov/pub/pmc/oa_pdf/1e/5d/CPZ1-2-0.PMC9522315.pdf","citationCount":"0","resultStr":null,"platform":"Semanticscholar","paperid":null,"PeriodicalName":"Current Protocols","FirstCategoryId":"1085","ListUrlMain":"https://doi.org/10.1002/cpz1.563","RegionNum":0,"RegionCategory":null,"ArticlePicture":[],"TitleCN":null,"AbstractTextCN":null,"PMCID":null,"EPubDate":"","PubModel":"","JCR":"","JCRName":"","Score":null,"Total":0}
引用次数: 0
Abstract
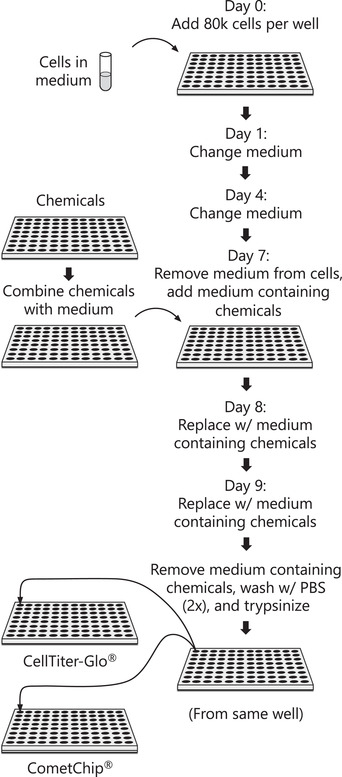
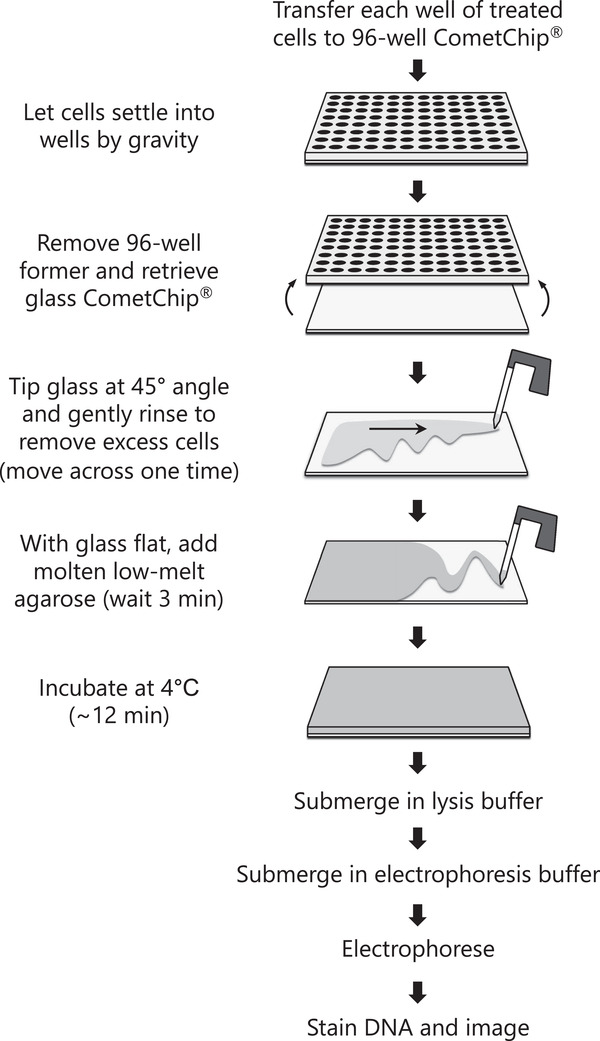
Exposure to DNA damaging agents can lead to mutations that cause cancer. The liver is particularly vulnerable because it contains high levels of Cytochrome P450 enzymes that can convert xenobiotics into DNA reactive metabolites that form potentially carcinogenic bulky DNA adducts. As such, current requirements for preclinical testing include in vivo testing for DNA damage in the liver, which often requires many animals. Given that efforts are underway in many countries to reduce or eliminate the use of animals in research, there is a critical need for fast and robust in vitro tests to discern whether xenobiotics or potential pharmaceutical agents can damage the hepatocyte genome. One possible approach is to leverage the alkaline comet assay, which is used to assess genotoxicity based on the ability of damaged DNA to become free to migrate toward the anode during electrophoresis. The comet assay, however, has several limitations. The assay is (i) slow and (ii) vulnerable to experimental noise, (iii) it is difficult to detect bulky DNA adducts since they do not directly affect DNA migration, and (iv) cell types typically used do not have robust metabolic capacity. To address some of these concerns, we have developed the "HepaCometChip" (a.k.a. the HepaRG CometChip), wherein metabolically competent cells are incorporated into a higher throughput CometChip platform. Repair trapping is used to increase sensitivity for bulky lesions: undetectable bulky lesions are converted into repair intermediates (specifically, single-strand breaks) that can be detected with the assay. Here, we describe a protocol for performing the HepaCometChip assay that includes handling and dosing of HepaRG cells and performing the CometChip assay. With its higher throughput, ability to capture metabolic activation, and sensitivity to bulky lesions, the HepaCometChip offers a potential alternative to the use of animals for genotoxicity testing. © 2022 The Authors. Current Protocols published by Wiley Periodicals LLC. Basic Protocol 1: HepaRG cell culturing and dosing Basic Protocol 2: CometChip assay.
使用HepaCometChip法进行广谱DNA损伤分析。
暴露在DNA破坏剂中会导致突变,从而导致癌症。肝脏尤其脆弱,因为它含有高水平的细胞色素P450酶,可以将异种生物转化为DNA反应性代谢物,形成可能致癌的大块DNA加合物。因此,目前对临床前测试的要求包括肝脏DNA损伤的体内测试,这通常需要许多动物。鉴于许多国家正在努力减少或消除在研究中使用动物,因此迫切需要进行快速和强有力的体外试验,以确定外源药物或潜在药物制剂是否会损害肝细胞基因组。一种可能的方法是利用碱性彗星测定法,该方法用于评估遗传毒性,该遗传毒性是基于电泳过程中受损DNA自由迁移到阳极的能力。然而,彗星试验有一些局限性。该分析(i)速度慢,(ii)易受实验噪声的影响,(iii)很难检测到大体积的DNA加合物,因为它们不直接影响DNA迁移,(iv)通常使用的细胞类型不具有强大的代谢能力。为了解决这些问题,我们开发了“HepaCometChip”(又名HepaRG CometChip),其中代谢能力强的细胞被纳入更高通量的CometChip平台。修复捕获用于增加对大体积病变的敏感性:无法检测到的大体积病变被转化为可以用该分析检测到的修复中间体(特别是单链断裂)。在这里,我们描述了一种执行HepaCometChip检测的方案,包括处理和给药HepaRG细胞和执行CometChip检测。HepaCometChip具有更高的通量、捕获代谢激活的能力和对大体积病变的敏感性,为动物遗传毒性测试提供了一种潜在的替代方法。©2022作者。Wiley期刊有限责任公司发布的当前方案。基本方案1:HepaRG细胞培养和给药基本方案2:CometChip试验。
本文章由计算机程序翻译,如有差异,请以英文原文为准。




 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


