通过异汀和烯酸盐的串联反应,以 TBD 为媒介非对映选择性地获得功能化的 3-烯基-2-吲哚
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
本发明公开了以 1,5,7-三氮杂双环[4.4.0]癸-5-烯为介导,通过容易获得的异汀和异烯酸酯与官能化 3-烯基-2-吲哚的串联反应。该反应可以在温和的反应条件下(32 个实例,收率高达 84%),以良好的收率和优异的官能团耐受性合成多种 3-烯基-2-氧化吲哚。目前的策略将为可持续合成功能化 3-烯基-2-吲哚衍生物提供一条新途径。我们还证明了 3-烯基-2-氧化吲哚作为关键起始原料(KSMs)的重要性,因为它们在合成具有药用价值的氧化吲哚添加吡唑、恶唑和香豆素杂交化合物方面具有实用价值。本文章由计算机程序翻译,如有差异,请以英文原文为准。
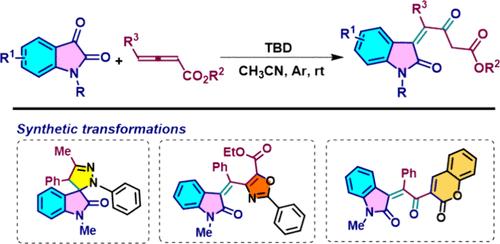
TBD-Mediated Diastereoselective Access to Functionalized 3-Alkenyl-2-oxindoles via the Tandem Reaction of Isatins and Allenoates
The 1,5,7-triazabicyclo[4.4.0]dec-5-ene–mediated tandem reaction of easily accessible isatins and allenoates to functionalized 3-alkenyl-2-oxindoles is disclosed. The reaction allows the synthesis of a wide range of 3-alkenyl-2-oxindoles in good yields with excellent functional group tolerance under mild reaction conditions (32 examples, up to 84% yields). The current strategy will provide a novel path for the sustainable synthesis of functionalized 3-alkenyl-2-oxindole derivatives. We have also demonstrated the significance of 3-alkenyl-2-oxindoles as key starting materials (KSMs) via their synthetic utility in producing oxindole-appended pyrazole, oxazole, and coumarin hybrids of medicinal relevance.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


