鉴定新型抗葡萄球菌命中化合物。
IF 2.1
4区 医学
Q3 BIOTECHNOLOGY & APPLIED MICROBIOLOGY
引用次数: 0
摘要
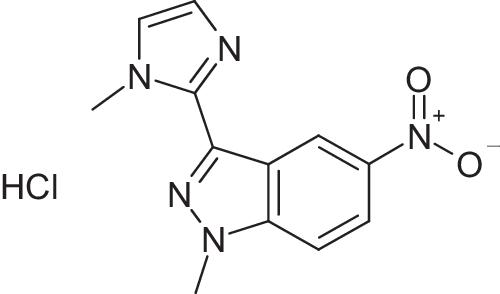
金黄色葡萄球菌是全球最常见的鼻腔生物膜形成病原体之一,对大多数抗生素都产生了耐药机制。因此,寻找具有未开发作用机制的新型抗葡萄球菌药物,特别是具有抗生物膜活性的药物,引起了人们极大的兴趣。丝氨酰-tRNA 合成酶被认为是开发抗菌药的一个有前景的药物靶点。我们将早先通过表型筛选发现的具有抗球菌活性的化合物与金黄色葡萄球菌 SerRS 的合成位点进行了分子对接,发现了 7 个具有低抑制活性的命中化合物。此外,我们还从以前测试过的对金黄色葡萄球菌 ThrRS 有抑制活性的化合物中寻找金黄色葡萄球菌 SerRS 抑制剂,这些化合物属于同一类氨基酰-tRNA 合成酶。在这些化合物中,我们发现了六个命中化合物。我们选择了四个化合物进行抗菌研究,发现活性最强的化合物 1-甲基-3-(1H-咪唑-1-甲基-2-基)-5-硝基-1H-吲唑对金黄色葡萄球菌耐多药临床分离株的 MIC 值介于 78.12 至 156.2 µg/ml 之间。但在抗生物膜研究中,该化合物出现沉淀。因此,我们使用了 3-[N'-(2-羟基-3-甲氧基亚苄基)肼基]-6-甲基-4H-[1,2,4]三嗪-5-酮进行抗生物膜研究。研究发现,该化合物对金黄色葡萄球菌 25923 的浮游生物和生物膜培养物的生长具有显著的抑制作用,其 MIC 值为 32 µg ml-1。同时,该化合物对大肠埃希氏菌 ATCC 47076 没有抗菌活性。因此,该化合物可作为对耐多药生物膜形成金黄色葡萄球菌分离物的有效杀菌剂。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Identification of novel antistaphylococcal hit compounds
Staphylococcus aureus is one of the most common nosocomial biofilm-forming pathogens worldwide that has developed resistance mechanisms against majority of the antibiotics. Therefore, the search of novel antistaphylococcal agents with unexploited mechanisms of action, especially with antibiofilm activity, is of great interest. Seryl-tRNA synthetase is recognized as a promising drug target for the development of antibacterials. We have carried out molecular docking of compounds with antistaphycoccal activity, which were earlier found by us using phenotypic screening, into synthetic site of S. aureus SerRS and found seven hit compounds with low inhibitory activity. Further, we have performed search of S. aureus SerRS inhibitors among compounds which were previously tested by us for inhibitory activity toward S. aureus ThrRS, that belong to the same class of aminoacyl-tRNA synthetases. Among them six hits were identified. We have selected four compounds for antibacterial study and found that the most active compound 1-methyl-3-(1H-imidazol-1-methyl-2-yl)-5-nitro-1H-indazole has MIC values toward S. aureus multidrug-resistant clinical isolates ranging from 78.12 to 156.2 µg/ml. However, this compound precipitated during anti-biofilm study. Therefore, we used 3-[N’-(2-hydroxy-3-methoxybenzylidene)hydrazino]-6-methyl-4H-[1,2,4]triazin-5-one with better solubility (ClogS value = 2.9) among investigated compounds toward SerRS for anti-biofilm study. It was found that this compound has a significant inhibitory effect on the growth of planktonic and biofilm culture of S. aureus 25923 with MIC value of 32 µg ml−1. At the same time, this compound does not reveal antibacterial activity toward Esherichia coli ATCC 47076. Therefore, this compound can be proposed as effective antiseptic toward multidrug-resistant biofilm-forming S. aureus isolates.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Antibiotics
医学-免疫学
CiteScore
6.60
自引率
3.00%
发文量
87
审稿时长
1 months
期刊介绍:
The Journal of Antibiotics seeks to promote research on antibiotics and related types of biologically active substances and publishes Articles, Review Articles, Brief Communication, Correspondence and other specially commissioned reports. The Journal of Antibiotics accepts papers on biochemical, chemical, microbiological and pharmacological studies. However, studies regarding human therapy do not fall under the journal’s scope. Contributions regarding recently discovered antibiotics and biologically active microbial products are particularly encouraged. Topics of particular interest within the journal''s scope include, but are not limited to, those listed below:
Discovery of new antibiotics and related types of biologically active substances
Production, isolation, characterization, structural elucidation, chemical synthesis and derivatization, biological activities, mechanisms of action, and structure-activity relationships of antibiotics and related types of biologically active substances
Biosynthesis, bioconversion, taxonomy and genetic studies on producing microorganisms, as well as improvement of production of antibiotics and related types of biologically active substances
Novel physical, chemical, biochemical, microbiological or pharmacological methods for detection, assay, determination, structural elucidation and evaluation of antibiotics and related types of biologically active substances
Newly found properties, mechanisms of action and resistance-development of antibiotics and related types of biologically active substances.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


