Can the Hock Process Be Used to Produce Phenol from Polystyrene?
IF 14.4
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
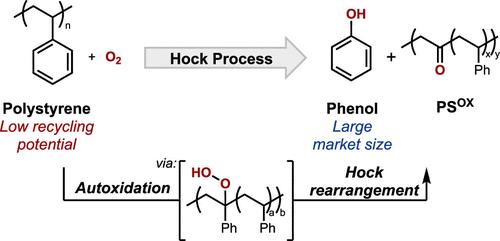
Polystyrene (PS) is a widely used thermoplastic polymer, but its very low recycling rate has motivated consideration of chemical conversion strategies to convert waste PS into value-added products. Oxidation methods have been widely studied, but they typically generate benzoic acid, a product with a relatively low market demand. Phenol is a higher volume chemical that would be an appealing target, but no methods currently exist for the conversion of PS into phenol. The repeat unit in PS closely resembles cumene, the primary feedstock used to produce phenol through the Hock process. Here, we investigate prospects for adapting the Hock process to PS, generating hydroperoxides through the autoxidation of benzylic C–H bonds followed by the acid-promoted rearrangement of the hydroperoxides to afford phenol and a partially oxygenated polymer. Experimental and computational studies of dimeric and trimeric PS model compounds show that neighboring phenyl rings impose conformational constraints that raise the barrier to hydrogen-atom transfer from the tertiary benzylic C–H bond. These effects are also evident with PS and contribute to lower yields of phenol when PS is subjected to Hock process conditions. These results provide valuable insights that have important implications for other efforts that seek to adapt small-molecule reactivity to polymeric feedstocks.
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
24.40
自引率
6.00%
发文量
2398
审稿时长
1.6 months
期刊介绍:
The flagship journal of the American Chemical Society, known as the Journal of the American Chemical Society (JACS), has been a prestigious publication since its establishment in 1879. It holds a preeminent position in the field of chemistry and related interdisciplinary sciences. JACS is committed to disseminating cutting-edge research papers, covering a wide range of topics, and encompasses approximately 19,000 pages of Articles, Communications, and Perspectives annually. With a weekly publication frequency, JACS plays a vital role in advancing the field of chemistry by providing essential research.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


