Protective effect of metformin on the NG-nitro-l-arginine methyl ester (l-NAME)-induced rat models of preeclampsia
IF 2.5
3区 生物学
Q3 BIOCHEMISTRY & MOLECULAR BIOLOGY
Biochemical and biophysical research communications
Pub Date : 2024-11-13
DOI:10.1016/j.bbrc.2024.150996
引用次数: 0
Abstract
Background
Preeclampsia (PE) is a complex multi-organ disorder characterized by systemic inflammation, endothelial dysfunction, and vasoconstriction, which manifests as hypertension, with or without proteinuria. Effective preventive strategies for PE are currently lacking in clinical practice, leading to significant morbidity and mortality among mothers and newborns.
Objective
This study aims to investigate the impact of metformin (MET) on the l-NAME-induced PE rat model, focusing on the mechanisms through which MET may exert its effects.
Methods
Thirty pregnant Sprague-Dawley (SD) rats were randomly assigned to three groups: Control, PE, and PE + MET, on gestational day 0 (GD0). Regularly measure blood pressure and 24-h proteinuria, and collect tissue samples on GD20. Enzyme-linked immunosorbent assay (ELISA) was used to analyze inflammatory factors, endothelial function biomarkers, angiogenic factors, and apoptosis-related factors in the rat plasma. Western blot, RT-qPCR, and immunohistochemistry techniques were employed to determine the expression levels of key apoptotic proteins in placental tissue.
Results
The study findings demonstrate that MET administration improves blood pressure and 24-h proteinuria, alleviates fetal growth restriction, ameliorate inflammation cytokines, and restores the balance of angiogenic factors and endothelial function. Moreover, MET inhibits the expression levels of critical apoptotic proteins in the plasma and placental tissue of PE-like rats.
Conclusion
The results suggest that MET shows promise in alleviating symptoms associated with l-NAME-induced PE in rats, preserving endothelial function, enhancing angiogenesis, reducing inflammation, inhibiting placental apoptosis, improving placental function, and promoting fetal growth. These findings highlight MET as a potential therapeutic agent for the prevention and treatment of preeclampsia.
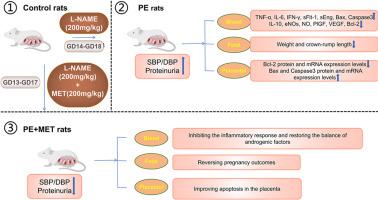
二甲双胍对 NG-硝基-精氨酸甲酯(l-NAME)诱导的子痫前期大鼠模型的保护作用。
背景:子痫前期(PE)是一种复杂的多器官疾病,以全身炎症、内皮功能障碍和血管收缩为特征,表现为高血压,伴有或不伴有蛋白尿。目前,临床上缺乏有效的 PE 预防策略,导致母亲和新生儿的发病率和死亡率显著上升:本研究旨在探讨二甲双胍(MET)对 l-NAME 诱导的 PE 大鼠模型的影响,重点是二甲双胍发挥其作用的机制:方法:将 30 只妊娠 Sprague-Dawley (SD) 大鼠随机分为三组,分别为对照组、PE 组和 PE + MET 组:方法:30 只妊娠 Sprague-Dawley (SD) 大鼠在妊娠第 0 天(GD0)被随机分为三组:对照组、PE 组和 PE + MET 组。定期测量血压和 24 小时蛋白尿,并在 GD20 时采集组织样本。使用酶联免疫吸附试验(ELISA)分析大鼠血浆中的炎症因子、内皮功能生物标志物、血管生成因子和细胞凋亡相关因子。研究采用了 Western 印迹、RT-qPCR 和免疫组化技术来确定胎盘组织中关键凋亡蛋白的表达水平:研究结果表明,服用 MET 可改善血压和 24 小时蛋白尿,缓解胎儿生长受限,改善炎症细胞因子,恢复血管生成因子的平衡和内皮功能。此外,MET还能抑制PE样大鼠血浆和胎盘组织中关键凋亡蛋白的表达水平:结果表明,MET有望缓解l-NAME诱导的PE大鼠的相关症状,保护内皮功能,增强血管生成,减轻炎症反应,抑制胎盘凋亡,改善胎盘功能,促进胎儿生长。这些研究结果突出表明,MET 是预防和治疗子痫前期的一种潜在治疗药物。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊
CiteScore
6.10
自引率
0.00%
发文量
1400
审稿时长
14 days
期刊介绍:
Biochemical and Biophysical Research Communications is the premier international journal devoted to the very rapid dissemination of timely and significant experimental results in diverse fields of biological research. The development of the "Breakthroughs and Views" section brings the minireview format to the journal, and issues often contain collections of special interest manuscripts. BBRC is published weekly (52 issues/year).Research Areas now include: Biochemistry; biophysics; cell biology; developmental biology; immunology
; molecular biology; neurobiology; plant biology and proteomics

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


