Key role of PPAR-γ-mediated suppression of the NFκB signaling pathway in rutin's antidepressant effect
IF 6.7
1区 医学
Q1 CHEMISTRY, MEDICINAL
引用次数: 0
Abstract
Background
Depression is a chronic and recurrent disorder with an unknown etiology. Efficacious antidepressant treatments with minimal side effects are urgently needed. Neuroinflammation may contribute to depression, as anti-inflammatory drugs have been shown to alleviate depressive symptoms in clinical practice. Rutin, a naturally occurring flavonoid derived from plants, is abundant in many antidepressant herbs, including Hemerocallis citrina Baroni. Historical Chinese medical texts, including the renowned Compendium of Materia Medica, document H. citrina Baroni as possessing antidepressant properties. Rutin, one of its primary active constituents, is recognized for its anti-inflammatory effects. Despite this, little is known about its specific target and mechanism.
Methods
In the present study, molecular docking, and surface plasmon resonance imaging (SPRi) analysis were used to identify the special targets of rutin. Meanwhile, the potential antidepressant effects were evaluated in the chronic social defeat stress (CSDS) paradigm, an animal model of depression. Then, Western blotting, quantitative real-time polymerase chain reaction (qRT-PCR), Co-immunoprecipitation (Co-IP) as well as antagonists of PPAR-γ were utilized to investigate the mechanism underlying the antidepressant effect of rutin.
Results
Both molecular docking and SPRi analysis showed high docking scores and interactions between rutin and PPAR-γ. In vivo, rutin promoted the nuclear translocation of PPAR-γ in the hippocampus of mice, inhibited NFκB-mediated inflammatory pathways, and subsequently reduced the expression of pro-inflammatory factors (e.g., iNOS, IL-6), aligning with an antidepressant effect. However, this therapeutic effect was attenuated by GW9662, a specific antagonist of PPAR-γ.
Conclusion
As a result of activating PPAR-γ and inhibiting NFκB pathway activation, rutin reduces neuroinflammation and exhibits an antidepressant effect. These findings shed light on the antidepressant mechanism of rutin and could be valuable for the development of new antidepressants.
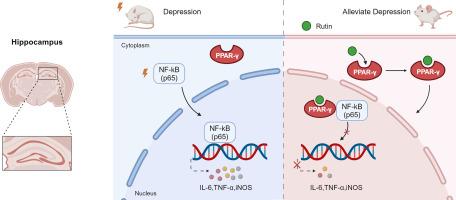
PPAR-γ介导的抑制 NFκB 信号通路在芦丁抗抑郁作用中的关键作用
背景介绍抑郁症是一种病因不明、反复发作的慢性疾病。目前迫切需要副作用小、疗效显著的抗抑郁治疗方法。神经炎症可能是导致抑郁症的原因之一,因为在临床实践中,抗炎药物已被证明可减轻抑郁症状。芦丁是从植物中提取的一种天然黄酮类化合物,在许多抗抑郁草药中含量丰富,包括 Hemerocallis citrina Baroni。历史上的中医典籍,包括著名的《本草纲目》,都记载了 Hemerocallis citrina Baroni 具有抗抑郁特性。芦丁是其主要活性成分之一,具有公认的消炎作用。尽管如此,人们对其特定靶点和机制知之甚少:本研究采用分子对接和表面等离子体共振成像(SPRi)分析来确定芦丁的特殊靶点。同时,在抑郁症动物模型--慢性社会挫败应激(CSDS)范式中评估了芦丁的潜在抗抑郁作用。然后,利用Western印迹、实时定量聚合酶链反应(qRT-PCR)、共免疫沉淀(Co-IP)以及PPAR-γ拮抗剂等方法研究芦丁的抗抑郁作用机制:结果:分子对接和SPRi分析均显示芦丁与PPAR-γ之间存在较高的对接得分和相互作用。在体内,芦丁促进了 PPAR-γ 在小鼠海马中的核转位,抑制了 NFκB 介导的炎症通路,继而降低了促炎因子(如 iNOS、IL-6)的表达,与抗抑郁作用相一致。然而,这种治疗效果被 PPAR-γ 的特异性拮抗剂 GW9662 削弱:结论:芦丁能激活 PPAR-γ 并抑制 NFκB 通路的激活,从而减少神经炎症并发挥抗抑郁作用。这些发现揭示了芦丁的抗抑郁机制,对开发新型抗抑郁药物具有重要价值。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Phytomedicine
医学-药学
CiteScore
10.30
自引率
5.10%
发文量
670
审稿时长
91 days
期刊介绍:
Phytomedicine is a therapy-oriented journal that publishes innovative studies on the efficacy, safety, quality, and mechanisms of action of specified plant extracts, phytopharmaceuticals, and their isolated constituents. This includes clinical, pharmacological, pharmacokinetic, and toxicological studies of herbal medicinal products, preparations, and purified compounds with defined and consistent quality, ensuring reproducible pharmacological activity. Founded in 1994, Phytomedicine aims to focus and stimulate research in this field and establish internationally accepted scientific standards for pharmacological studies, proof of clinical efficacy, and safety of phytomedicines.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


