One-pot sequential synthesis of unsymmetrical diarylmethanes using methylene chloride as a C1-synthon
IF 2.9
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
Bisindolylmethane (BIM) and its derivatives are widely used in the pharmaceutical industry due to their significant biological activities. However, most reported synthetic methods are focused on the synthesis of symmetric BIMs, while the synthesis of unsymmetrical BIMs remains a challenge. Herein, an unprecedented two-step one-pot method to afford unsymmetrically substituted 3,3′-BIM frameworks, using methylene chloride (DCM) as the C1-synthon is reported. In this protocol, the formation of two C–C bonds can be achieved via a one-pot reaction. The utility of commercially available phenols and anilines was also demonstrated in the construction of unsymmetrical diarylmethanes. This protocol provides a straightforward approach to access diverse unsymmetrical diarylmethane derivatives under simple and mild conditions. The broad substrate compatibility and good functional group tolerance of the protocol support its practical application potential.
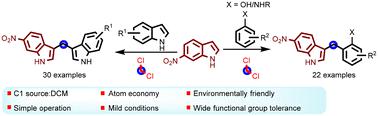
以二氯甲烷为 C1-质子,单锅顺序合成不对称的二芳基甲烷
双吲哚甲烷(BIM)及其衍生物因其显著的生物活性而被广泛应用于制药业。然而,大多数报道的合成方法都集中在对称 BIM 的合成上,而非对称 BIM 的合成仍然是一个挑战。本文报告了一种前所未有的两步一步法,以二氯甲烷 (DCM) 作为 C1-合成锍,制备非对称取代的 3,3′-BIM 框架。在该方案中,可通过一次反应形成两个 C-C 键。在非对称二芳基甲烷的构建过程中,也证明了市售苯酚和苯胺的实用性。该方案提供了一种在简单温和的条件下获得各种非对称二芳基甲烷衍生物的直接方法。该方案具有广泛的底物兼容性和良好的官能团耐受性,因而具有实际应用潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
The international home of synthetic, physical and biomolecular organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


